In this segment, we examine the 12 most common endocrine disruptors according to the Environmental Working Group and 4 other common environmental chemicals we come in contact with daily and review the potential health effects of these compounds according to the most recent research. We review the evidence showing how these compounds may be contributing to various hormonal and reproductive disorders, cognitive and neurological disorders, kidney and liver disease, immunological disorders and cancer. We also make recommendations on how you can protect you and your family against exposure to each of these toxic compounds.
In this segment, we examine the 12 most common endocrine disruptors according to the Environmental Working Group and 4 other common environmental chemicals we come in contact with daily and review the potential health effects of these compounds according to the most recent research. We review the evidence showing how these compounds may be contributing to various hormonal and reproductive disorders, cognitive and neurological disorders, kidney and liver disease, immunological disorders and cancer. We also make recommendations on how you can protect you and your family against exposure to each of these toxic compounds.
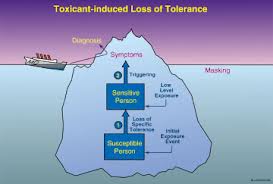 In the next segment, we introduce the concept of chemical tolerance and body burden. We introduce the concept of toxicant-induced loss of tolerance (TILT) and examine how this develops and why this may be important for more and more people today. We review common signs and symptoms of loss of chemical tolerance including chemical sensitivity to a wide variety of chemicals in your environment.
In the next segment, we introduce the concept of chemical tolerance and body burden. We introduce the concept of toxicant-induced loss of tolerance (TILT) and examine how this develops and why this may be important for more and more people today. We review common signs and symptoms of loss of chemical tolerance including chemical sensitivity to a wide variety of chemicals in your environment.
Key concepts for this issue
- What are “Endocrine Disruptors”?
- 12 Common Endocrine Disruptors as Identified by the Environmental Working Group
- What are “Xenoestrogens”?
- 4 Other Notable Environmental Chemicals that you Probably Come in Contact with on a Daily Basis
What are “Endocrine Disruptors”?
From the moment of conception, human growth and development is regulated by tiny amounts of biochemicals called hormones. Our bodies manufacture trace quantities of these chemical messengers, which enter our bloodstream and t ravel throughout our circulatory system, managing our metabolism and controlling the function of specific tissues and organs. Hormones are able to produce an array of sophisticated regulatory signals crucial to human health at part per billion or part per trillion levels in the blood. Disruption of the human hormone (endocrine) system can cause a broad range of illnesses, including reproductive and metabolic disorders and cancer. There are numerous environmental chemicals that we potentially come in contact with on a daily basis that have been shown to disrupt the endocrine system. Chemicals that disrupt the endocrine system are called endocrine disruptors.
ravel throughout our circulatory system, managing our metabolism and controlling the function of specific tissues and organs. Hormones are able to produce an array of sophisticated regulatory signals crucial to human health at part per billion or part per trillion levels in the blood. Disruption of the human hormone (endocrine) system can cause a broad range of illnesses, including reproductive and metabolic disorders and cancer. There are numerous environmental chemicals that we potentially come in contact with on a daily basis that have been shown to disrupt the endocrine system. Chemicals that disrupt the endocrine system are called endocrine disruptors.
12 Common Endocrine Disruptors as Identified by the EWG
In October, 2013, the Environmental Working Group (EWG) published a list of 12 common environmental chemicals and toxins that act as endocrine disruptors in human physiology.[1] This means they disrupt hormonal signaling (alter hormonal responses) in the human body. Since hormones act as biochemical messengers and regulators of physiologic function, this can lead to a wide range of physiological problems and it’s interesting to note that most of them impact multiple endocrine organs simultaneously.
 Here is a list of those chemicals:
Here is a list of those chemicals:
- BPA
- Dioxin
- Atrazine
- Phthalates
- Perchlorate
- Fire retardants
- Lead
- Arsenic
- Mercury
- Perfluorinated chemicals
- Organophosphate pesticides
- Glycol Ethers
As we did in the previous list of hazardous and toxic compounds, we will take a close look at the most current research on each one of these compounds (not already covered in the last article) and discuss impacts of exposure on human health.
 1. Bisphenol A (BPA)
1. Bisphenol A (BPA)
Bisphenol A (BPA) is a plastics chemical currently used in enormous amounts to manufacture hard plastic water bottles and to make epoxy linings of metal food cans, like those for canned infant formula. Another common source of BPA is cash register and credit card receipts, the handling of which have recently been shown to cause elevations in BPA in the blood, as just published by the prestigious Journal of the American Medical Association a couple months ago.[2]
How common is BPA exposure?
Although its long-time use in consumer products has come with assurances of its safety from industry, studies conducted over the past 20 years now show it to be not only a ubiquitous pollutant in the human body–it contaminates approximately 93% of the population according to the CDC [3]—but also a potent developmental toxin at very low doses. One recent study that examined over 80 published human biomonitoring studies that measured BPA concentrations in human tissues, urine, blood, and other fluids, concluded the following on BPA exposure:
“Available data from biomonitoring studies clearly indicate that the general population is exposed to BPA and is at risk from internal exposure to unconjugated BPA.”[4]
 What are the health effects of BPA exposure?
What are the health effects of BPA exposure?
In September, 2008, the National Toxicology Program of NIH determined that BPA may pose risks to human development, raising concerns for early puberty, prostate effects, breast cancer, and behavioral impacts from early-life exposures.[5] But this came after many warnings from consumer environmental groups such as the Environmental Working Group and an accumulation of studies spanning more than 10 years showing that BPA had potentially harmful effects in the human body.[6]
A recent review of the most current literature on the health effects of BPA published in December 2013, concluded:
“The growing human literature correlating environmental BPA exposure to adverse effects in humans, along with laboratory studies in many species including primates, provides increasing support that environmental BPA exposure can be harmful to humans, especially in regards to behavioral and other effects in children.”[7]
 What are “xenoestrogens”?
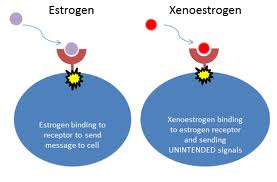
What are “xenoestrogens”?
BPA is currently understood to be a xenoestrogen. Xenoestrogens are a type of foreign or synthetic compound (“xeno” means foreign) that imitates estrogen in the human body. This means they stimulate estrogen receptors and produce an estrogenic effect on a living organism even though they differ chemically from the natural estrogenic hormones produced internally by the endocrine system. Synthetic xenoestrogens are widely used industrial compounds, which include BPA, PCBs, and phthalates, among others. Xenoestrogens are clinically significant because they can mimic the effects of endogenous (naturally-produced) estrogen and thus have been implicated in several types of cancer, including breast cancer [8-14], prostate cancer [15-18], and testicular cancer.[19,20]
 Extensive literature has raised concerns about their impacts on human development, particularly during the developmental states of the brain and nervous system and endocrine system during pregnancy and implications for many endocrine-related disorders such as metabolic disorder, diabetes and obesity [21-29], cardiovascular diseases [30-34], liver and kidney toxicity [35-38], abnormal prostate development [39-42], low sperm count [43-45] and other reproductive disorders.[46-52]
Extensive literature has raised concerns about their impacts on human development, particularly during the developmental states of the brain and nervous system and endocrine system during pregnancy and implications for many endocrine-related disorders such as metabolic disorder, diabetes and obesity [21-29], cardiovascular diseases [30-34], liver and kidney toxicity [35-38], abnormal prostate development [39-42], low sperm count [43-45] and other reproductive disorders.[46-52]
“Importantly, many of these studies have reported BPA-related effects at very low doses, e.g., at exposure levels less than 50 μg kg−1 day−1, the current “safe” dose that is considered acceptable for daily intake by the U.S. Food and Drug Administration (FDA) and that is also the reference dose established by the U.S. Environmental Protection Agency. This suggests that, like endogenous hormones, environmental estrogens can elicit biological effects at extremely low concentrations.”[53]
 Who are the most vulnerable to the effects of BPA?
Who are the most vulnerable to the effects of BPA?
Many studies indicate that pregnant women and the developing fetus and baby are the most vulnerable to BPA’s toxic effects.[54-59] Unfortunately, they also have the highest BPA exposure of any age group. The federal Food and Drug Administration (FDA) estimates that babies have 12.5 times more BPA exposure than adults.[60] Therefore, it is critical for pregnant mothers to avoid BPA as much as possible.
Additionally, a large number of studies have linked BPA exposure with neural development and neurological disorders [61-70], learning and behavioral disorders [71-74] and abnormal growth and development.[75,76]
As one recent study concluded regarding the current safety standards for BPA:
 “We propose that a new risk assessment for BPA is needed based on a) the extensive new literature reporting adverse effects in animals at doses below the current reference dose; b) the high rate of leaching of BPA from food and beverage containers, leading to widespread human exposure; c) reports that the median BPA level in human blood and tissues, including in human fetal blood, is higher than the level that causes adverse effects in mice; and d) recent epidemiologic evidence that BPA is related to disease in women.”[77]
“We propose that a new risk assessment for BPA is needed based on a) the extensive new literature reporting adverse effects in animals at doses below the current reference dose; b) the high rate of leaching of BPA from food and beverage containers, leading to widespread human exposure; c) reports that the median BPA level in human blood and tissues, including in human fetal blood, is higher than the level that causes adverse effects in mice; and d) recent epidemiologic evidence that BPA is related to disease in women.”[77]
 How to Avoid Exposure: Go fresh instead of canned – many food cans are lined with BPA – or research which companies don’t use BPA or similar chemicals in their products. Use powdered baby formulas instead of canned. Don’t use polycarbonate plastics for warm food or drinks. Say no to receipts, since thermal paper is often coated with BPA. Avoid plastics marked with a “PC” for polycarbonate or recycling label #7. For more tips, check out: www.ewg.org/bpa
How to Avoid Exposure: Go fresh instead of canned – many food cans are lined with BPA – or research which companies don’t use BPA or similar chemicals in their products. Use powdered baby formulas instead of canned. Don’t use polycarbonate plastics for warm food or drinks. Say no to receipts, since thermal paper is often coated with BPA. Avoid plastics marked with a “PC” for polycarbonate or recycling label #7. For more tips, check out: www.ewg.org/bpa
Avoiding BPA for Pregnant Mothers and Infants
 Avoid BPA in formula and baby bottles
Avoid BPA in formula and baby bottles
BPA exposure comes primarily from canned foods and polycarbonate food containers, but baby bottles and baby formula can also contain BPA. Most manufacturers now make BPA-free baby bottles. Glass is the safest and most durable option. Never microwave baby food or drinks in plastic containers. Many parents who have replaced their polycarbonate baby bottles are unaware that BPA contaminates liquid baby formula sold in metal cans. A 1990s test by FDA show BPA leaching from metal cans into all brands of liquid formula. Since formula can make up to 100% of a baby’s diet over her first 6 months of life, parents unable to breast feed should choose a powdered formula if your baby tolerates it. Powdered formula appears to be BPA-free.[78]
Almost all canned foods sold in the United States have a BPA-based epoxy liner that leaches BPA into the food. EWG tested 97 canned foods and found detectable levels of BPA in more than half of the foods. The highest concentrations were in canned meats, pasta and soups. Pregnant women and children should limit their consumption of canned foods to avoid BPA. Rinsing canned fruit or vegetables may reduce the amount of BPA you ingest.
 Avoid BPA in water and food containers
Avoid BPA in water and food containers
Less BPA leaches from plastic water bottles and food containers than from cans into canned foods and baby formula. Nevertheless, it is good to take simple precautions to reduce your exposure. Polycarbonate plastics are rigid, transparent and used for food storage containers and water bottles, among other things. Trace amounts of BPA can migrate from these containers, particularly if used for hot food or liquids. Soft or cloudy-colored plastic does not contain BPA.
Avoid putting any plastic containers in microwaves and do not heat up food in plastic containers. Some metal water bottles lined with an epoxy-based enamel coating could leach BPA. Look for stainless steel bottles that do not have a liner.[79]
 2. Dioxin
2. Dioxin
Dioxins are major environmental pollutants and are of concern because of their highly toxic potential. They have the dubious distinction of belonging to the “dirty dozen”–a group of dangerous chemicals known as persistent organic pollutants (POPs). In the 1970s, dioxin was identified as a contaminant in Agent Orange, the notorious defoliant deployed by the U.S. during the Vietnam War and blamed for diabetes and other diseases among exposed personnel.[80] Based on animal data and on human epidemiology data, dioxin is classified by IARC as a “known human carcinogen” although the EPA has categorized dioxin as a “likely human carcinogen”.
The name “dioxins” is often used for the family of structurally and chemically related polychlorinated dibenzo para dioxins (PCDDs) and polychlorinated dibenzofurans (PCDFs). Certain dioxin-like polychlorinated biphenyls (PCBs) with similar toxic properties are also included under the term “dioxins”. Dioxins are found throughout the world in the environment and they accumulate in the food chain, mainly in the fatty tissue of animals, including fish. More than 90% of human exposure is through food, mainly meat and dairy products, fish and shellfish.[81]
 Sources of dioxin contamination
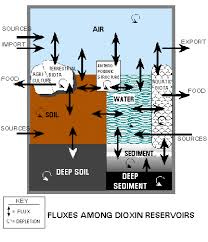
Sources of dioxin contamination
Dioxins are mainly by-products of industrial processes but can also result from natural processes, such as volcanic eruptions and forest fires. Dioxins are unwanted by-products of a wide range of manufacturing processes including smelting, chlorine bleaching of paper pulp and the manufacturing of some herbicides and pesticides. In terms of dioxin release into the environment, uncontrolled waste incinerators (solid waste and hospital waste) are often the worst culprits, due to incomplete burning. Dioxins are found throughout the world in the environment. The highest levels of these compounds are found in some soils, sediments and food, especially dairy products, meat, fish and shellfish. Very low levels are found in plants, water and air.[82]
Accumulation of dioxins in animals and the human body
Once dioxins have entered the body, they endure a long time because of their chemical stability and their ability to be absorbed by fat tissue, where they are then stored in the body. Their half-life in the body is estimated to be seven to eleven years! In the environment, dioxins tend to accumulate in the food chain. The higher in the animal food chain one goes, the higher the concentration of dioxins. Due to the omnipresence of dioxins, all people have background exposure which contributes to their body burden.[83]
 Infant exposure to dioxins
Infant exposure to dioxins
Again, infants are exposed to the highest amounts and are most vulnerable to exposure. EWG research found that the amount of dioxin a nursing infant ingests daily is up to 77 times higher than the level EPA has proposed to protect the endocrine and immune systems.[84] The fact that both breast milk and formula are contaminated with dioxin highlights the urgent need for EPA to finish its assessment on dioxins.
Health effects of dioxin exposure
Short-term exposure of humans to high levels of dioxins may result in skin lesions, such as chloracne and patchy darkening of the skin, and altered liver function.[85] Long-term exposure is linked to impairment of the developing immune system [86-92], respiratory system [93-99], nervous system [100-107], endocrine system [108-114] and reproductive functions [115-124] and cause cancer.[125-134] For cancer risk, the situation is concerning: the general public is exposed to up to 1,200 times more dioxin than regulatory agencies typically consider safe.[135]
 The developing fetus, infants and other vulnerable subgroups
The developing fetus, infants and other vulnerable subgroups
The developing fetus is most sensitive to dioxin exposure. The newborn, with rapidly developing organ systems, may also be more vulnerable to certain effects. Some individuals or groups of individuals may be exposed to higher levels of dioxins because of their diets (e.g., high consumers of fish in certain parts of the world) or their occupations (e.g., workers in the pulp and paper industry, in incineration plants and at hazardous waste sites, to name just a few).[136]
How to Avoid Exposure: Eat organic foods whenever possible. Products including meat, fish, milk, eggs and butter are most likely to be contaminated, but you can cut down on your exposure by eating fewer animal products and eating organic foods when possible. Trimming fat from meat and consuming low fat dairy products may decrease the exposure to dioxin compounds. Make sure your fish oil supplements are checked for dioxin levels.
 3. Atrazine
3. Atrazine
Atrazine, the most heavily used herbicide in the United States but banned in the European Union, is a cancer-causing weed killer applied to 50 million acres of corn each year. After it is applied each spring, it runs off cornfields and through drinking water plants into the tap water of millions of Mid-western homes. While many larger water utilities suppress atrazine levels through special treatments, toxic traces remain in finished water.[137] Atrazine has gained the attention of a number of current researchers.
According to amphibian endocrinologist Tyrone Hayes of U.C. Berkeley, exposure to the herbicide Atrazine results in what amounts to chemical castration in frogs.[138] In an interview with Minnesota Public Radio news, Hayes said:
 “One of the things we discovered is that atrazine chemically castrates the frog, meaning that it causes a reduction in the male hormone, testosterone, which results in things like decreased sperm count, a decrease in the voice box which controls the male’s ability to attract mates.”
“One of the things we discovered is that atrazine chemically castrates the frog, meaning that it causes a reduction in the male hormone, testosterone, which results in things like decreased sperm count, a decrease in the voice box which controls the male’s ability to attract mates.”
But Hayes says the enzyme that atrazine activates in frogs is the same one found in humans. It converts testosterone into estrogen.
 “This same enzyme, or machinery if you will, controls estrogen production in humans. And atrazine is associated in laboratory rodents with development of mammary cancer, or breast cancer, and there are epidemiological studies that associate atrazine exposure to breast cancer in humans.”[139]
“This same enzyme, or machinery if you will, controls estrogen production in humans. And atrazine is associated in laboratory rodents with development of mammary cancer, or breast cancer, and there are epidemiological studies that associate atrazine exposure to breast cancer in humans.”[139]
Atrazine has been linked to delayed puberty and prostate inflammation in animals, and some research has linked it to prostate cancer in men with possible implications for other reproductive cancers.[140-142]
 Atrazine Continues to Contaminate Surface Water and Drinking Water in the United States
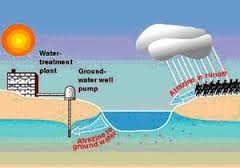
Atrazine Continues to Contaminate Surface Water and Drinking Water in the United States
The most recent data confirms that atrazine continues to contaminate watersheds and drinking water across the Midwest and Southern United States. Atrazine was found in 80% of drinking water samples taken in 153 public water systems in 2007 to 2008. All twenty watersheds sampled during this time had detectable levels of atrazine, and sixteen had average concentrations above the level that has been shown to harm plants and wildlife.[143]
How to Avoid Exposure: Eat organic whenever possible! Drink filtered water certified to remove atrazine. Check out the EWG’s “Shopper’s Guide to Pesticides in Produce” for more information: http://www.ewg.org/foodnews/guide.php?key=40442072
 4. Phthalates
4. Phthalates
Phthalates (pronounced “tha–lates”) are a group of chemicals used to soften and increase the flexibility of plastic and vinyl. Polyvinyl chloride (PVC) is made softer and more flexible by the addition of phthalates. Phthalates are used in hundreds of consumer products, including cosmetics and personal care products, perfume, hair spray, soap, shampoo, nail polish, and skin moisturizers. They are used in consumer products such as plastic and vinyl baby toys, shower curtains, wallpaper, vinyl miniblinds, food packaging, and plastic wrap. Phthalates are also used in wood finishes, detergents, adhesives, plastic plumbing pipes, lubricants, medical tubing and fluid bags, solvents, insecticides, medical devices, building materials, and vinyl flooring.[144]
 How are people exposed to phthalates?
How are people exposed to phthalates?
You can be exposed to low levels of phthalates through air, water, or food or if you use cosmetics, personal care products, cleaning products, or other plastic and vinyl products that contain them. Exposure to low levels of phthalates may come from eating food packaged in plastic that contains phthalates or breathing dust in rooms with vinyl miniblinds, wallpaper, or recently installed flooring that contain phthalates. You could also be exposed by drinking water that contains phthalates, though it is not known how common that is. Children can be exposed to phthalates by chewing on soft vinyl toys or other products made with them. Children can also be exposed by breathing household dust that contains phthalates or using IV tubing or other medical devices made with phthalates.[145]
 How much environmental phthalate gets into our bodies?
How much environmental phthalate gets into our bodies?
In September, 2000, scientists from the Centers for Disease Control and Prevention (CDC) conducted the first accurate measurements of human phthalate exposures, and reported finding phthalates in every one of 289 people tested, at unexpectedly high levels.[146] Levels of some phthalates in U.S. women of childbearing age have been found to exceed the government’s safe levels set to protect against birth defects, according to another CDC study.[147] Results of phthalate testing in more than 2,500 people ages 6 and above confirmed the CDC’s original findings: phthalate exposures are widespread across the population, and women are exposed at higher levels than men.[148] In a 2007 study of girls age 6 to 8 spearheaded by Mount Sinai School of Medicine, phthalates were found in every one of 90 girls tested.[149] Phthalates are widespread contaminants in the environment as well [150,151).
 What are the health effects of phthalate exposure?
What are the health effects of phthalate exposure?
Epidemiological studies of ordinary people have linked high phthalate levels to a wide variety of problems with the male reproductive tract, including reduced sperm motility and concentration, increased damage to sperm DNA, alterations in hormone levels in adult men.[152-154] Exposure to phthalates in the developing fetus and infant boys (through the mother’s breast milk) has been associated with congenital anomalies, decreased testosterone levels and infertility[155-158] which confirmed earlier animal studies.[159-160]
Recent studies indicate phthalates may produce non-reproductive health effects in both males and females.[161,162] Phthalate exposure has been associated with a wide range of health effects, including allergies, asthma and atopic dermatitis (eczema) [163-166], pulmonary function [167], alterations in thyroid function [168-171], learning, memory and behavioral problems in children [172-176] and breast cancer.[177-181]
 How to Avoid Exposure: Avoid plastic food containers, children’s toys (some phthalates are already banned in kids’ products), and plastic wrap made from PVC, which has the recycling label #3. Some personal care products also contain phthalates, so read the labels and avoid products that simply list added “fragrance,” since this catch-all term sometimes means hidden phthalates. Find phthalate-free personal care products with EWG’s Skin Deep Database: www.ewg.org/skindeep/
How to Avoid Exposure: Avoid plastic food containers, children’s toys (some phthalates are already banned in kids’ products), and plastic wrap made from PVC, which has the recycling label #3. Some personal care products also contain phthalates, so read the labels and avoid products that simply list added “fragrance,” since this catch-all term sometimes means hidden phthalates. Find phthalate-free personal care products with EWG’s Skin Deep Database: www.ewg.org/skindeep/
5. Perchlorate
Perchlorate is the explosive chemical of solid rocket and missile fuel that is produced for and used in rocket fuel, fireworks, ammunition and explosives. The National Resources Defense Council (NRDC) estimates it contaminates the drinking water of more than 20 million Americans.[182] Perchlorate contaminates more than 350 drinking water sources in California alone! Nationally, perchlorate contamination of drinking water has been confirmed by testing in 22 states.[183] Perchlorate contamination of food is also widespread.
 Perchlorate in our milk and food supply
Perchlorate in our milk and food supply
A 2008 Environmental Working Group (EWG) analysis of FDA data shows that three quarters of 285 commonly consumed foods and beverages are contaminated with perchlorate. That means that every day, the average two-year-old is exposed to more than half of the EPA “safe” dose of perchlorate from food alone. This is especially troubling in the 22 states that also are exposed to perchlorate through contaminated tap water.[184]
A 2004 analysis by EWG found that milk from cows raised in some parts of California may expose infants and children to more of a toxic rocket fuel chemical than is considered safe by the U.S. Environmental Protection Agency (EPA). In their study which looked for perchlorate in California supermarket milk, EWG found perchlorate in almost every sample tested: 31 out of 32 samples purchased from grocery stores in Los Angeles and Orange counties. EWG also obtained results of tests for perchlorate in milk by the California Department of Food and Agriculture (CDFA), which found perchlorate in all 32 samples of milk collected from unspecified sources in Alameda, Sacramento, and San Joaquin counties.[185]
 Our children our most vulnerable to perchlorate exposure
Our children our most vulnerable to perchlorate exposure
EWG’s computer-assisted analysis of federal dietary data shows that by drinking milk contaminated with the levels of perchlorate found in the two studies, 7 percent of women of childbearing age would get a daily dose of rocket fuel larger than the level currently considered safe by the EPA. But children are by far more at risk: Half of all children 1 to 5 would exceed EPA’s provisional daily safe dose just by drinking milk, and more than a third would get twice that dose. One-third of children 6 to 11 would get a larger dose than EPA says is safe, with one-fifth consuming twice as much. These risk estimates assume zero perchlorate exposure from other sources, such as contaminated tap water and foods.[186]
 Sources of perchlorate
Sources of perchlorate
Perchlorate occurs both naturally and as a manufactured compound. Natural deposits tend to be of low concentration, except for the high natural perchlorate concentrations found in Chile.[187] Man-made perchlorate sources can be many times more concentrated than most natural sources. Their production and use can lead to releases of perchlorate into the environment. Sites that have been identified with relatively high concentrations of perchlorate contamination involve manufacturing, testing, or disposal of solid rocket propellant; manufacturing of perchlorate compounds; and industrial manufacturing operations where perchlorate compounds were used as reagents.
 How does perchlorate get into our food?
How does perchlorate get into our food?
Contamination of perchlorate occurs in the soil and water. Many crop and feed plants, including lettuce, cucumbers, tomatoes and alfalfa, concentrate perchlorate in their tissues when grown with contaminated water. This perchlorate, in turn, can be passed along in cow (or human) milk. A study published in 2009 reported elevated perchlorate levels in powdered infant formula! They also observed that infants given formula reconstituted with perchlorate-contaminated drinking water would be consuming unsafe amounts of the chemical, according to the current EPA standards.[188]
 What are the health effects of perchlorate exposure?
What are the health effects of perchlorate exposure?
Perchlorate can affect the thyroid gland’s ability to make essential hormones.[189,190] For fetuses, infants and children, disruptions in thyroid hormone levels have been shown to cause lowered IQ, mental retardation, loss of hearing and speech, and motor skill deficits.[191-196]
In addition, toddlers are particularly vulnerable because they eat and drink substantial amounts of food and water relative to their small size. Also affected by perchlorate levels are women with low iodine levels, which may account for up to 1/3 of women in the U.S.[197,198]
How to Avoid Exposure: You can reduce perchlorate in your drinking water by installing a reverse osmosis filter or buying filtered water. (You can get help finding one at: www.ewg.org/report/ewgs-water-filter-buying-guide) As for food, it’s almost impossible to avoid perchlorate, but you can reduce exposure by buying organic food, especially milk, and making sure you are getting enough iodine in your diet. Eating iodized salt is one good way.
 6. Fire retardants
6. Fire retardants
Fire retardants are found in many types of furniture, computers, televisions and other electronics in the home and office. Fire retardants are commonly found in polyurethane foam furniture like couches and upholstered chairs, mattresses and pads, futons and carpet padding and even children’s pajamas. Product studies also find fire retardants in the foam of children’s products including car seats, changing table pads, portable crib mattresses, nap mats, and nursing pillows. Foam products made before 2005 are most hazardous. Older foam items commonly contain PBDEs (polybrominated diphenyl ethers), a highly toxic fire retardant mixture now removed from the U.S. market. Since 2004 these chemicals have not been manufactured in the U.S. However once PBDEs were withdrawn, other concerning chemicals took their place.[199]
 How do fire retardants affect health?
How do fire retardants affect health?
According to a review published in a scientific journal just a couple months ago:
“Plausible outcomes associated with brominated flame retardant (BFR) exposure include diabetes, neurobehavioral and developmental disorders, cancer, reproductive health effects and alteration in thyroid function.”[200]
In rodents, repeated exposure to PBDEs results in thyroid hormone disruption, developmental neurotoxicity, some changes of fetal development, and hepatotoxic effects.[201]
Very serious health effects are associated with exposure to PBDEs, but toxicity depends on the compound and the amount that one is exposed to. PBDEs are chemically similar to PCBs and, not surprisingly, they show similar health effects as well. They are biomagnified toxic compounds, meaning they accumulate within food chains. Brominated fire retardants are still being studied for health effects but so far have been found to have the following effects in human studies:
 Thyroid disruption
Thyroid disruption
According to an article published in 2005, brominated fire retardants have a striking structural similarity with the thyroid hormones and may constitute a potential health risk by interfering with thyroid hormone homeostasis. The thyroid hormones are essential for normal brain development. Disruption of the hormonal balance may lead to serious and permanent defects of neurological functioning. Brominated flame retardants may interfere with thyroid synthesis, transport, receptor binding, and elimination. The clinical consequences have so far not been firmly established, but results from animal studies suggest that even subtle disturbances of thyroid homeostasis during pregnancy may have serious implications for the developing brain.[202-205]
 Endocrine disruption
Endocrine disruption
Many studies have found some type of endocrine disruption that occurs on exposure to fire retardants. One of the mechanisms that has been identified is that this compound acts as a xenoestrogen in the body as discussed earlier in this article. Xenoestrogens are foreign compounds that mimic estrogen in the body. The potential health impacts of xenoestrogens are still not completely understood.[206-208]
 Neurobehavioral dysfunction
Neurobehavioral dysfunction
Studies have shown that prenatal exposure to organohalogens, including brominated flame retardants, influences motor, cognitive, and behavioral performance in school-age children.[209, 210] Numerous laboratory studies on mice have shown them to have neurotoxic or neurobehavioral effects[211-213] and in rodents the neurological impacts are more significant if exposure occurs during a specified period of time during neurological development.[214]
Studies have shown an association between fire retardant exposure and reproductive problems such as spontaneous abortion and abnormal uterine tissue and affecting male genitourinary conditions as well.[215-217],
 Cardiac and metabolic abnormalities
Cardiac and metabolic abnormalities
Studies have also shown associations with fire retardant exposure and metabolic syndrome in the U.S. and effects on the heart in animal studies.[218, 219]
Cancer
The International Agency for Research on Cancer (IARC) has determined that PBDEs are possibly carcinogenic to humans.[220]
 Exposure to PBDEs in breast milk
Exposure to PBDEs in breast milk
Exposure to PBDEs is nearly impossible to avoid due to their ubiquity in the air, water, food, human fat, and breast milk. The fetus is exposed to the toxins in utero as well as from breast milk, which is where the toxins are transferred from the mother to the baby. A Swedish study published in 2006 found that the concentration of PBDEs in breast milk doubled every five years during the 25-year period that was examined in the study[221] while another showed that higher levels of PBDEs were found in breast milk of women in the U.S. than their European counterparts.[222]
 How common is exposure to fire retardants?
How common is exposure to fire retardants?
Like PCBs, their long-banned chemical relatives, the brominated fire retardants are persistent in the environment and bioaccumulative, meaning they build up in people’s bodies over a lifetime. There are ubiquitous in our environment, meaning they are found everywhere. As one newspaper writer puts it: “PBDEs: They are everywhere, they accumulate and they spread”.[223] In 2004, the Environmental Working Group (EWG) performed the first nationwide tests for brominated fire retardants in house dust. This study found unexpectedly high levels of these neurotoxic chemicals in every home sampled.[224] EWG’s test results indicate that consumer products, not industrial releases, are the most likely sources of the rapid buildup of PBDEs in people, animals and the environment, which has been documented by tests from Europe to the Arctic.
 Scientists now recognize that indoor environmental contamination, including contaminants accumulating in household dust, pose a substantial health risk to the population.[225] It is no surprise that American homes are contaminated with PBDEs since these fire retardants are added to thousands of everyday products, including computers, cars, TVs, furniture and clothing. But the EWG study showed the surprising degree to which these chemicals are escaping into the air from consumer products and exposing people, especially children, who continually ingest or inhale dust.
Scientists now recognize that indoor environmental contamination, including contaminants accumulating in household dust, pose a substantial health risk to the population.[225] It is no surprise that American homes are contaminated with PBDEs since these fire retardants are added to thousands of everyday products, including computers, cars, TVs, furniture and clothing. But the EWG study showed the surprising degree to which these chemicals are escaping into the air from consumer products and exposing people, especially children, who continually ingest or inhale dust.
How to Avoid Exposure: “Remove old foam (pre-2005) from your home. Clean the dust in your home frequently and regularly, especially if you have infants or children at home.”
7. Lead (previously discussed in last segment)
8. Arsenic (previously discussed in last segment)
9. Mercury (previously discussed in last segment)
10. Perfluorinated chemicals (PFCs)
 What are PFCs?
What are PFCs?
PFCs are a large group of manufactured compounds that are widely used to make everyday products more resistant to stains, grease, and water. For example, PFCs may be used to keep food from sticking to cookware, to make sofas and carpets resistant to stains, to make clothes and mattresses more waterproof, and may also be used in some food packaging, as well as in some firefighting materials. Because they help reduce friction, they are also used in a variety of other industries, including aerospace, automotive, building and construction, and electronics.[226]
PFCs in the environment
PFCs break down very slowly in the environment and are considered persistent organic pollutants (POPs). There is widespread wildlife and human exposure to several PFCs, including perfluorooctanoic acid (PFOA) and perfluorooctane sulfonate (PFOS). Perfluorochemicals are so widespread and extraordinarily persistent that studies show approximately 99% of Americans have these chemicals in their bodies.[227]
 Where do they come from?
Where do they come from?
Both PFOA and PFOS are byproducts of other commercial products, meaning they are released into the environment when other products are made, used, or discarded. PFOS is no longer manufactured in the United States, and PFOA production has been reduced and will soon be eliminated. More research is needed to fully understand all sources of human exposure, but people are most likely exposed to these compounds by consuming PFC-contaminated water or food, or by using products that contain PFCs.
Are PFCs like other toxins?
Unlike many other persistent environmental chemicals, PFCs are not stored in body fat. However, PFCs are similar to other persistent chemicals, because the half-life, or the amount of time it takes for 50% of the chemical to leave the human body, for some of these chemicals, is several years. This slow elimination time makes it difficult to determine how changes in lifestyle, diet, or other exposure-related factors influence blood levels.
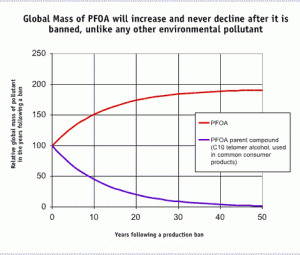 One particularly notorious compound called PFOA has been shown to be “completely resistant to biodegradation.” In other words, PFOA doesn’t break down in the environment – ever. That means that even though the chemical was banned after decades of use, it will be showing up in people’s bodies for countless generations to come. This is worrisome, since PFOA exposure has been linked to decreased sperm quality, low birth weight, kidney disease, thyroid disease and diabetes, among other health issues.[228]
One particularly notorious compound called PFOA has been shown to be “completely resistant to biodegradation.” In other words, PFOA doesn’t break down in the environment – ever. That means that even though the chemical was banned after decades of use, it will be showing up in people’s bodies for countless generations to come. This is worrisome, since PFOA exposure has been linked to decreased sperm quality, low birth weight, kidney disease, thyroid disease and diabetes, among other health issues.[228]
Let’s take a quick look at some of this research on the health effects of exposure to PFCs:
 Endocrine disruption
Endocrine disruption
PFCs are a known endocrine disruptor. Many observational studies suggest that endocrine disruptors contribute to cancer, diabetes, obesity, metabolic syndrome, and infertility.[229] Until recently, most of this data was derived from animal studies but there is growing epidemiologic health data that support associations between adult exposures and outcomes. Recent studies suggest that there are several mechanisms by which PFCs such as PFOA may lead to endocrine disruption.[230]
Studies indicate there is disruption in both male and female reproductive physiology.[231] In males, there have been associations with decreased sperm count, semen quality and reproductive hormones.[232] Endocrine disruptors have also been implicated in the development of obesity.[233]
Thyroid disruption
The thyroid-disrupting effects of PFCs have been documented in many studies, and disruption has been shown to occur with total T3, total T4, and free T4 in the U.S. general population.[234-237]
 Reduced immune function
Reduced immune function
There is evidence from both epidemiology and laboratory studies that PFCs may be immunotoxic, affecting both cell-mediated and humoral immunity.[238] PFC exposure has also been associated with allergic skin reactions such as atopic dermatitis and pediatric atopy.[239]
Multiple organ and tissue effects
PFCs can cause adverse effects on multiple organs, including the liver and pancreas and have been shown to cause damage at multiple biological levels including oxidative stress, DNA damage, membrane instability, suppressed filtration rate, and reduced body weight.[240] PFCs have also been associated with osteoarthritis in women.[241]
 Neurological and neurobehavioral effects
Neurological and neurobehavioral effects
PFCs have been shown to be neurotoxic to the brain[242] and cause fetal growth and developmental problems and affect birth outcomes.[243-247]
How to Avoid Exposure: Skip non-stick pans as well as stain and water-resistant coatings on clothing, furniture and carpets. Don’t do the Scotchguard®! Stay away from Teflon®, Goretex® or any foreign-sounding names to protect you from stains and water!
 11. Organophosphate pesticides
11. Organophosphate pesticides
“In this century, several hundred billion pounds of pesticides have been produced and released into the global environment. Nearly 5 billion pounds of the insectide DDT alone have been applied both indoors and out since it was introduced in 1939, and DDT is one of nearly 600 pesticides currently registered for use in the world. By 1969, almost 60,000 different products were sold containing some combination of pesticides along with their inert ingredients. As we approach the 21st century, an additional 5 to 6 billion pounds of insecticides, herbicides, fungicides, rodenticides and other biocides are added to the world’s environment each year, with roughly ¼ of this amount released or sold in the U.S.”[248]
 Pesticides can be extremely toxic to human health and the environment. U.S. and international government agencies alike have linked pesticides to nervous system toxicity, cancer, hormone system disruption and IQ deficits among children. Studies indicate that children are the most vulnerable to the damaging effects of pesticides. According to one expert on childhood exposure to pesticides:
Pesticides can be extremely toxic to human health and the environment. U.S. and international government agencies alike have linked pesticides to nervous system toxicity, cancer, hormone system disruption and IQ deficits among children. Studies indicate that children are the most vulnerable to the damaging effects of pesticides. According to one expert on childhood exposure to pesticides:
“Children are especially vulnerable to health damage from pesticides. Following conception, a child’s susceptibility changes as organ systems grow and certain functions mature, such as the detoxification potential of the liver or the filtration potential of the kidneys. Children may be especially vulnerable to carcinogens during periods when their cells are normally reproducing most rapidly, generally between conception and age five. They may be more susceptible to loss of brain function if exposed to neurotoxins during critical periods of development.”[249]
 Neurological effects on children from pesticide exposure
Neurological effects on children from pesticide exposure
Studies show even small amounts of these chemicals add up and can impair a child’s neurological development when they are exposed during the early, critical stages of their development. Numerous studies have been conducted in humans and animals to evaluate the potential effect of myriad exposures, including environmental contaminants, on neurologic development.[250] Neurodevelopmental deficits include a broad spectrum of disorders and dysfunctions such as autism spectrum disorder, attention deficit hyperactivity disorder (ADHD), decreased intelligence, learning disabilities, developmental delays, emotional or behavioral problems, and deficits in gross or fine motor skills. The exact prevalence of these deficits is difficult to ascertain; however, it has been estimated that approximately 3 to 8% of infants and 12% of children are affected by one or more of these conditions.[251] Strong arguments have been made that our government agencies and the science on which policies are based have been insufficient to protect our children against these harmful chemicals.[252]
 Pregnancy and fetal exposure to pesticides
Pregnancy and fetal exposure to pesticides
Epidemiological data shows that prenatal exposure to OP pesticides may be associated with an increased risk of pervasive developmental disorders, delays in cognitive development, and attentional deficits. In children, OP exposure has been associated with behavioral problems, poorer short-term memory and motor skills, and longer reaction time.[253]
One recent study concluded the following:
“These findings support the notion that prenatal exposure to pesticides—at levels not producing adverse health outcomes in the mother—can cause lasting adverse effects on brain development in children. Pesticide exposure therefore may contribute to a “silent pandemic” of developmental neurotoxicity.”[254]
 Three epidemiological studies published in April, 2011, show a clear link between a mother’s exposure to organophosphate (OP) insecticides during pregnancy and deficits to children’s learning and memory that persist through the ages of 6 to 9. (255)
Three epidemiological studies published in April, 2011, show a clear link between a mother’s exposure to organophosphate (OP) insecticides during pregnancy and deficits to children’s learning and memory that persist through the ages of 6 to 9. (255)
“The risks of dietary exposures to synthetic pesticides, especially organophosphate and pyrethroid insecticides, are greatest during pregnancy and childhood, when the brain and nervous system are most vulnerable. These are two groups that should really avoid eating foods with high levels of pesticide residues.” (256)
In an award-winning study published by the journal Environmental Science & Technology in 2009, Payne-Sturges of the EPA’s National Center for Environmental Research analyzed CDC biomonitoring data for organophosphate insecticide exposure in American children. She concluded that two in five American children are exposed to enough organophosphate pesticides to exceed safe amounts![257]
 In May 2010, a team led by Maryse F. Bouchard of the Harvard School of Public Health analyzed the same biomonitoring data and found that the children with elevated organophosphate exposure were more likely to be diagnosed with attention deficit-hyperactive disorder.[258] Several studies have also shown that agricultural workers have substantially greater opportunity for pesticide exposure than the population at large.[259-262]
In May 2010, a team led by Maryse F. Bouchard of the Harvard School of Public Health analyzed the same biomonitoring data and found that the children with elevated organophosphate exposure were more likely to be diagnosed with attention deficit-hyperactive disorder.[258] Several studies have also shown that agricultural workers have substantially greater opportunity for pesticide exposure than the population at large.[259-262]
“Pesticides, while designed specifically to kill certain organisms, are also associated with a host of very serious health problems in people, including neurological deficits, ADHD, endocrine system disruption and cancer. My advice to consumers is to whenever possible avoid exposure to pesticides, including pesticide residues on food.”
–Andrew Weil, MD, Founder and Director, Arizona Center for Integrative Medicine[263]
 How to Avoid Exposure: Buy organic produce. Consumers can markedly reduce their intake of pesticide residues and their exposure to antibiotic-resistant bacteria by choosing organic produce and meat, according to researchers at Stanford University who reviewed a massive body of scientific studies on the much-debated issue.[264]
How to Avoid Exposure: Buy organic produce. Consumers can markedly reduce their intake of pesticide residues and their exposure to antibiotic-resistant bacteria by choosing organic produce and meat, according to researchers at Stanford University who reviewed a massive body of scientific studies on the much-debated issue.[264]
“Consumers selecting organic produce ingest fewer pesticides. They also eat meats that harbor fewer deadly bacteria. While you still need to take responsibility for eating a varied and healthy diet, you can rest assured that organic food provides a healthier choice for people and the planet.”[265]
Use EWG’s Shopper’s Guide to Pesticides in Produce, which can help you find the fruits and vegetables that have the fewest pesticide residues at: www.ewg.org/foodnews/
 12. Glycol Ethers
12. Glycol Ethers
Glycol ethers (GE) are a family of solvents, the use of which has increased dramatically since the 1960’s, in domestic and occupational settings and persist mainly in water-based products, such as paints, inks, varnishes and cleaning agents. There are two subgroups: E series and P series. EGME, among the E series, is the reference substance for most studies. Human and animal studies have shown that glycol ethers can cause adverse reproductive, developmental, and hematological effects through inhalation, dermal absorption, and ingestion. The reproductive organs, nervous system, blood and blood-forming organs are the primary organs affected in acute and chronic intoxications of glycol ethers.[266]
 Congenital birth defects
Congenital birth defects
Studies in multiple countries show that glycol ethers act as human teratogens (substances that cause birth defects) when fetuses are exposed to these chemicals in utero.[267-269]
Fertility and reproductive health
Glycol ethers are a reprotoxic (toxic to the reproductive system) chemical causing infertility and genital atrophy on both sexes (shrinks the testicles and ovaries) and embryofetal toxicity through maternal exposure (malformation, growth alterations and functional deficits).[270-275] Studies show that glycol ether exposure is related to low motile sperm count in men.[276, 277] Female workers exposed to ethylene glycol in the semiconductor manufacturing industry have been shown to have prolonged menstrual cycles, inhibition of ovulation and prolonged time to pregnancy.[278-281]
Hematological effects
Studies show that glycol ethers are a hematological toxin in rat studies and leads to anemia in workers exposed to this toxic compound.[282-284]
 Neurologic effects
Neurologic effects
Overexposure to propylene glycol monomethyl ether (PGME) has been associated with increases in central nervous system depression in rat toxicology studies.[285] It is known that ethylene glycol ethers (EGEs) cross the blood-brain barrier and acute intoxication with these compounds causes disturbances of motor coordination, headaches, permanent cognitive dysfunction, central nervous system (CNS) depression and occasionally convulsions in humans [286].
 A recent rat study found that 4-week administration of a mixture of two ethylene glycol ethers (EGEs) decreased total antioxidant capacity and enhanced lipid peroxidation (oxidation of lipids) in the frontal cortex and hippocampus of the rats. These are the same mechanisms involved in neurodegenerative diseases, such as Alzheimer’s disease and Parkinson’s disease. The study concluded:
A recent rat study found that 4-week administration of a mixture of two ethylene glycol ethers (EGEs) decreased total antioxidant capacity and enhanced lipid peroxidation (oxidation of lipids) in the frontal cortex and hippocampus of the rats. These are the same mechanisms involved in neurodegenerative diseases, such as Alzheimer’s disease and Parkinson’s disease. The study concluded:
“The obtained results suggested that EGEs exerted adverse effects on the central nervous system (CNS) cells and may contribute in pathogenesis of neurodegenerative disorders.”[287]
How to Avoid Exposure: Start by checking out EWG’s Guide to Healthy Cleaning (www.ewg.org/guides/cleaners/) and avoid products with ingredients such as 2-butoxyethanol (EGBE) and methoxydiglycol (DEGME).
4 Other Notable Environmental Chemicals that You Should Avoid
1. Perchloroethylene/
Tetrachlorethylene (PERC) 
Perchloroethylene, also known as tetrachloroethylene, PCE, or PERC, is a chlorinated solvent long used in dry cleaning, industrial cleaning and production of other chemicals and consumer products. Biomonitoring surveys have detected it in the bodies of a significant number of Americans. It has been found in drinking water across the country. The federal Environmental Protection Agency has declared tetrachloroethylene, or PERC, a chemical used by many dry cleaners, a “likely human carcinogen.”[288]
 Dry cleaners use dangerous chemical solvents that can stick to clothing. Most cleaners in the U.S. still use PERC as a solvent for dry cleaning. A recent study by scientists at Georgetown University, published in 2011, in the journal Environmental Toxicology and Chemistry, found high levels of residual PERC on dry-cleaned wool, cotton and polyester (what do you have on now?). Subsequent dry cleaning cycles intensified these concentrations.[289] When inhaled, even low concentrations of PERC are known to cause respiratory and eye irritation, headache, dizziness and vision problems. Once in the environment, the chemical breaks down into other known human carcinogens, including trichloroethylene (TCE) and vinyl chloride.
Dry cleaners use dangerous chemical solvents that can stick to clothing. Most cleaners in the U.S. still use PERC as a solvent for dry cleaning. A recent study by scientists at Georgetown University, published in 2011, in the journal Environmental Toxicology and Chemistry, found high levels of residual PERC on dry-cleaned wool, cotton and polyester (what do you have on now?). Subsequent dry cleaning cycles intensified these concentrations.[289] When inhaled, even low concentrations of PERC are known to cause respiratory and eye irritation, headache, dizziness and vision problems. Once in the environment, the chemical breaks down into other known human carcinogens, including trichloroethylene (TCE) and vinyl chloride.
Health effects of PERCs
Based on toxicology, metabolism, animal studies, and human studies, occupational exposure to chlorinated aliphatic solvents (PERCs) has been associated with numerous adverse health effects, including central nervous system, reproductive, liver, and kidney toxicity, and carcinogenicity (initiation of cancer).[290]
 Cancer
Cancer
Human exposure to PERCs can produce various types of cancer, including esophageal cancer, cervical cancer, non-Hodgkin’s lymphoma, urinary bladder cancer, kidney cancer, leukemia and multiple myeloma (bone marrow cancer).[291-295]
Neurotoxicity and mental illness
PERC exposure has been found to be toxic to the brain and nervous system and lead to neurobehavioral problems.[296, 297] Studies have also linked prenatal and early childhood exposure to PERC-contaminated drinking water to mental illness, including bipolar disorder and PTSD.[298]
 Birth defects
Birth defects
A 2009 study also linked exposure to PERC-contaminated drinking water to birth defects.[299]
How to Avoid Exposure: Ask your cleaners what solvents they use. Watch out for perchloroethylene under any of its names (PERC, PCE, tetrachloroethylene), siloxane and hydrocarbon solvents. Patronize cleaners that use liquid carbon dioxide or the wet-cleaning method. Tetrachloroethylene can remain on clothes and evaporate into the air at home, unnecessarily exposing you and your family. Don’t leave dry cleaned items in the car for a long time because PERC vapors can build up inside the vehicle. Air dry cleaned clothes outdoors.[300]
 2. Formaldehyde
2. Formaldehyde
Formaldehyde is used in a vast array of industrial and consumer products, and there is widespread agreement that consumer exposures must be reduced to protect health.[301] It exists all around us in our homes, office buildings and automobiles and newer construction and material means increased exposure. According to ICIS, an international chemical market research firm, nearly two-thirds of the formaldehyde market is for resins to make construction materials such as plywood, particle board, fiber board, laminate flooring, carpeting, foam underlays, insulation and for vehicle coatings and brake linings. Other major uses include plastics for electronic, automotive and consumer goods, polyurethane foam, and adhesives and sealants for construction and consumer goods, including children’s toys. It is also used as the wet-strength resin added to paper products such as facial tissue, table napkins, and paper towels.
 Formaldehyde is also a precursor to polyfunctional alcohols which are used to make paints and explosives. The textile industry uses formaldehyde-based resins as finishers to make fabrics crease-resistant.[302a] Formaldehyde-based materials are key to the manufacture of automobiles, and used to make components for the transmission, electrical system, engine block, door panels, axles and brake shoes. There are also compounds known as “formaldehyde releasers” found in cosmetics and personal care products such as shampoos and conditioners which are known to release formaldehyde into these products, even those intended for children.[302b] The value of sales of formaldehyde and derivative products was over $145 billion in 2003, about 1.2% of the gross domestic product (GDP) of the United States and Canada. Including indirect employment, over 4 million people work in the formaldehyde industry across approximately 11,900 plants in the U.S. and Canada.[303]
Formaldehyde is also a precursor to polyfunctional alcohols which are used to make paints and explosives. The textile industry uses formaldehyde-based resins as finishers to make fabrics crease-resistant.[302a] Formaldehyde-based materials are key to the manufacture of automobiles, and used to make components for the transmission, electrical system, engine block, door panels, axles and brake shoes. There are also compounds known as “formaldehyde releasers” found in cosmetics and personal care products such as shampoos and conditioners which are known to release formaldehyde into these products, even those intended for children.[302b] The value of sales of formaldehyde and derivative products was over $145 billion in 2003, about 1.2% of the gross domestic product (GDP) of the United States and Canada. Including indirect employment, over 4 million people work in the formaldehyde industry across approximately 11,900 plants in the U.S. and Canada.[303]
 Health Effects of Formaldehyde Exposure
Health Effects of Formaldehyde Exposure
Acute (short-term) and chronic (long-term) exposures have been shown to cause respiratory symptoms and irritation to the skin, eyes, nose, throat and respiratory tract. Human studies have suggested an association between formaldehyde exposure and various cancers, including lung and nasopharyngeal cancer.[304] In 2011, the U.S. National Toxicology Program described formaldehyde as “known to be a human carcinogen“.[305] But it has also been shown to be toxic to the brain and nervous system, white blood cells (immune system), the lungs, the reproductive system and human DNA.[306] Let’s take a closer look at the various health effects linked to formaldehyde exposure:
 Effects Due to Acute Exposure
Effects Due to Acute Exposure
Formaldehyde is known to induce acute poisoning and cause irritation, as well as other immunotoxic effects. It is a highly reactive chemical that readily reacts with biological tissues, particularly the mucous tissues lining the respiratory tract and the eyes.[307,308] Acute mucus membrane irritation is the most common adverse effect of formaldehyde exposure, often leading to dry skin, dermatitis, tearing eyes, sneezing, and coughing.[309-312] Skin sensitization and dermal allergic reactions following dermal exposure to formaldehyde has been well documented.[313-318] Short exposures to high level formaldehyde are identified to cause a sudden onset of asthmatic symptoms called reactive airways dysfunction syndrome (RADS).[319-321] It was reported that the likelihood for the development of allergic asthma increases proportionately with level of indoor formaldehyde concentration, especially when levels exceed 0.08 ppm.[321-327]
Effects Due to Chronic Exposure
 Upper and Lower Respiratory Tract Toxicity
Upper and Lower Respiratory Tract Toxicity
Long-term exposure to elevated levels of formaldehyde, especially in the occupational setting, has been designated as the cause of irritation and pain such as upper and lower airway irritation, eye irritation, degenerative diseases, coughing, wheezing, body sores, chest pain, abdominal pain, and loss of appetite.[328-329] Long-term occupational formaldehyde exposure is also reported to be responsible for such chronic health effects as inflammatory and hyperplastic changes of the nasal mucosa, pharyngeal congestion, chronic pharyngitis, chronic rhinitis and loss of olfactory (sense of smell) function.[330-334]
 Neurotoxicity
Neurotoxicity
Chronic exposure to formaldehyde can be responsible for the symptoms of neurasthenia which include headaches, dizziness, sleep disorders, and memory loss. Many reports indicate that chronic exposure to formaldehyde increased the chances of headache and dizziness by 30%–60%.[335-337] Formaldehyde appears to have neurotoxic characteristics with systemic toxic effects. It is thus hypothesized that inhalation of formaldehyde, during the early postnatal period, can cause some neurological diseases with aging.[338]
 It was also recognized that apart from age and gender, environmental tobacco smoking (ETS) is perhaps the most consistent nongenetic risk factor for amyotrophic lateral sclerosis (ALS).[339, 340] (Recall that formaldehyde is an ingredient of tobacco.) ALS is a fatal, neurodegenerative disease caused by the degeneration of motor neurons system that controls voluntary muscle movement. In an investigation conducted by the American Cancer Society’s Cancer Prevention Study II, more than 1 million individuals were examined over time. Based on this study, it was concluded that individuals who reported formaldehyde exposure in the workplace (e.g., beauticians, pharmacists, morticians, chemists, laboratory technicians, physicians, veterinarians, dentists, firefighters, photographers, printers, and nurses) had a 34% higher rate of ALS than the non-exposure group.[341]
It was also recognized that apart from age and gender, environmental tobacco smoking (ETS) is perhaps the most consistent nongenetic risk factor for amyotrophic lateral sclerosis (ALS).[339, 340] (Recall that formaldehyde is an ingredient of tobacco.) ALS is a fatal, neurodegenerative disease caused by the degeneration of motor neurons system that controls voluntary muscle movement. In an investigation conducted by the American Cancer Society’s Cancer Prevention Study II, more than 1 million individuals were examined over time. Based on this study, it was concluded that individuals who reported formaldehyde exposure in the workplace (e.g., beauticians, pharmacists, morticians, chemists, laboratory technicians, physicians, veterinarians, dentists, firefighters, photographers, printers, and nurses) had a 34% higher rate of ALS than the non-exposure group.[341]
 Pulmonary Function Damage
Pulmonary Function Damage
In repetitive human exposure under occupational (or residential) conditions, formaldehyde has led to symptoms associated with irritation of the upper-respiratory tract and decreased pulmonary function.[342-347]
Hematotoxicity
Hematotoxicity is defined as toxicity to the blood and hematopoietic (blood-forming) system caused by chemical exposure, often resulting in decreased blood cell counts. It was demonstrated that long-term exposure of formaldehyde can decrease the number of white blood cells (an important part of your immune system).[348-350] A report by Huang et al. revealed that a previously healthy woman experienced lower than normal counts of white blood cells, red blood cells, platelet, and hemoglobin, just after 3 months of moving into a newly remodeled apartment.[351, 352]
 Reproductive Toxicity
Reproductive Toxicity
The potential role of formaldehyde as a teratogen (substance that causes birth defects) and its impacts on human reproduction are still a matter of scientific controversy. Until recently, very limited research has been conducted to specifically address such aspects of formaldehyde. However, a few studies found menstrual disorder and dysmenorrhea in women who were occupationally exposed to formaldehyde.[354, 355] In a case-control study, a significant association between spontaneous abortion and formaldehyde exposure was found in Finnish women who worked in pathology or histology laboratories for more than 3 days per week.[356] Another study focusing on female wood workers reported significantly decreased frequency of pregnancies in women exposed to high formaldehyde levels.[357]
 Genotoxicity (Toxicity to the DNA)
Genotoxicity (Toxicity to the DNA)
It was reported that formaldehyde exposure can induce DNA and chromosomal damage in human peripheral blood cells.[358, 359] A line of evidence indicated that formaldehyde is capable of directly reacting with DNA and producing genotoxic effects on portal-of-entry tissues, especially after exceeding liver biotransformation capacities.[360, 361] Chinese workers exposed to formaldehyde showed an increase in DNA damage in peripheral lymphocytes, when measured by single cell gel electrophoresis.[362-367]
 Carcinogenesis
Carcinogenesis
Based on comprehensive research and large-scale human studies conducted internationally, the International Agency for Research on Cancer (IARC) classified formaldehyde as a human carcinogen that can cause nasopharyngeal cancer.[368] According to this classification, formaldehyde is “a probable human carcinogen” under conditions of unusually high or prolonged exposure.[369] The U.S. National Toxicology Program (NTP) reported formaldehyde as a “known human carcinogen” in its 12th Report on Carcinogens.[370] There is sufficient evidence for a linkage between formaldehyde exposure and nasopharyngeal cancer, nasal and paranasal cancer, and leukemias.[371-373] Increased incidences of leukemia have also been reported in several occupational epidemiologic studies.[374-375]
 How to Avoid Exposure: You cannot avoid formaldehyde exposure completely but there are some steps you can take to avoid unnecessary exposure through products such as cosmetics and skin products. Check the label carefully, both for the ingredient formaldehyde itself and for the seven common “formaldehyde releasers” listed on the following EWG website: http://www.ewg.org/research/exposing-cosmetics-cover/formaldehyde-releasers
How to Avoid Exposure: You cannot avoid formaldehyde exposure completely but there are some steps you can take to avoid unnecessary exposure through products such as cosmetics and skin products. Check the label carefully, both for the ingredient formaldehyde itself and for the seven common “formaldehyde releasers” listed on the following EWG website: http://www.ewg.org/research/exposing-cosmetics-cover/formaldehyde-releasers
Or let EWG do the research for you. Check out the products in EWG’s Skin Deep Cosmetics Database (http://www.ewg.org/skindeep/). It contains information on more than 77,000 products and their ingredients. This database highlights toxic substances like formaldehyde — and those insidious formaldehyde releasers.
 3. Parabens
3. Parabens
Parabens are a group of artificial preservatives commonly found in many different cosmetics and body care products which have been in use since the 1920s. Parabens are used to prevent the growth of bacteria and mold in cosmetic products, increasing its shelf life. Studies suggest parabens can cause skin disorders and allergic reactions and act as endocrine disruptors in the human body and have been linked to breast cancer.[376] In 2006, the Centers for Disease Control (CDC) tested urine samples from a group of 100 adults and found parabens in nearly all samples.[377] Cosmetics typically contain mixtures of parabens. Five different parabens are fairly common in cosmetics: methyl-, ethyl-, propyl-, isopropyl-, and butylparaben (measured in combination with isobutylparaben); benzylparaben is less common.[378]
 How are we exposed to parabens?
How are we exposed to parabens?
Parabens are present in a wide variety of cosmetics and body care products, including moisturizers, face and skin cleansers, shampoos, conditioners, sunscreens, deodorants and antiperspirants, shaving gels, toothpastes, makeup, and many others. They are commonly used in sunscreens, in particular. They are also used as preservatives in food and beverages, and in some medications. Daily use of body care products can result in repeated, direct exposure to parabens. Parabens have also been detected in household dust.[379]
What are the potential health effects of paraben exposure?
Studies show that parabens can cause skin disorders and allergic reactions, act as endocrine disruptors in the human body in both males and females, are toxic to human DNA and have been linked to breast cancer.[380, 381] Let’s take a closer look at this research:
Skin and allergic reactions
Parabens have been shown to irritate the skin, causing atopic dermatitis and allergic skin reactions.[382-386]
 Endocrine disruption
Endocrine disruption
A wide range of in vitro and in vivo laboratory studies indicate that parabens are estrogenic, meaning they can mimic the hormone estrogen, and act as endocrine disruptors, disrupting normal function of the hormone system.[387-395] A recent study by scientists at the Harvard School of Public Health linked one type of paraben to impaired fertility in women.[396] A study published just this month showed that parabens cross the placenta and enter the fetal circulation of pregnant mothers.[397]
 Breast cancer
Breast cancer
There is growing concern in the scientific literature that paraben exposure can increase breast cancer risk.[398-402] In a 2004 study, traces of 5 different parabens were found in the breast cancer tumors of 19 of 20 women examined with methylbarapen (commonly found in sunscreens, deodorants and cosmetics) representing 62% of the total paraben recovered in the extractions.[403] More recent lab studies have since shown that parabens increase proliferation of breast cancer cells.[404-407] A recent study published only two months ago showed that in vitro, parabens can influence not only proliferation but also migratory and invasive properties of human breast cancer cells.[408]
Have these chemicals been regulated?
The European Union banned use of sodium methylparaben in fragrance because it can strip skin of pigment (SCCPNFP 1999, 2000). While FDA limits the levels of parabens allowed in foods and beverages, it does not regulate these chemicals in cosmetics and body care products.[409]
 How to Avoid Exposure: Because parabens are found in many everyday products, some exposure may be unavoidable. One way to reduce exposure is to switch to paraben-free cosmetics and body care products. Johnson & Johnson agreed to stop using most parabens in 2012, but they can still be found in numerous cosmetics. Read the labels carefully to spot products that contain parabens, especially the long-chained varieties—propylparaben, isopropylparaben, butylparaben and isobutylparaben.[410]
How to Avoid Exposure: Because parabens are found in many everyday products, some exposure may be unavoidable. One way to reduce exposure is to switch to paraben-free cosmetics and body care products. Johnson & Johnson agreed to stop using most parabens in 2012, but they can still be found in numerous cosmetics. Read the labels carefully to spot products that contain parabens, especially the long-chained varieties—propylparaben, isopropylparaben, butylparaben and isobutylparaben.[410]
 4. Triclosan
4. Triclosan
Triclosan is an artificial antimicrobial chemical used to kill bacteria on the skin and other surfaces. It is used in a variety of cosmetics and everyday products. Triclosan is the most common active ingredient in “antibacterial” liquid hand soaps, and is also found in toothpastes, deodorants, face and body washes, and acne treatments, among others. Besides cosmetics, triclosan is found in an increasing number of everyday items, such as dishwashing detergent, kitchen tools, toys, trash bags, bedding, bathmats, socks, and footwear.[411] However, a scientific advisory panel to the U.S. Food and Drug Administration (FDA) determined that “antibacterial” soaps, like those containing triclosan, were no better than regular soap and water at killing germs or reducing the spread of infection.[412] The American Medical Association recommends that triclosan and other “antibacterial” products not be used in the home, as they may promote antibiotic resistance in bacteria, because while repeated use kills most bacteria, this can allow resistant strains to flourish.[413]
 How common is triclosan exposure?
How common is triclosan exposure?
Widespread use has led to detection of triclosan in the breast milk, blood, and urine of ordinary people.[414-419] It was found in the urine of 61 percent of 90 girls ages 6 to 8 tested in a 2007 study by Mount Sinai School of Medicine.[420] Urine samples of triclosan have recently been shown to be reliable indicators of exposure.[421] In 2002, scientists found triclosan in 58 percent of 85 streams across the U.S.[422], most likely from discharges of treated wastewater. The amount of triclosan in the wastewater stream is estimated to be as much as 3 to 5 milligrams per person per day from residences alone [423]; in addition, substantial discharges of this antimicrobial agent are expected from laundries, hair salons, medical facilities, and many other commercial and industrial sites. Optimal water treatment can result in degradation and removal of 95 percent of triclosan [424]; however, small amounts may pass through the treatment plants to receiving waters.[425] Triclosan also has been found to contaminate household dust.[426] CDC’s national survey indicates that triclosan contaminates the bodies of 75% of the American population.[427]
 What are the potential health effects of triclosan exposure?
What are the potential health effects of triclosan exposure?
Although the majority of triclosan is eliminated from the blood through the urine, triclosan can accumulate in the liver and fatty tissue and build up in the bodies of people and animals over time. However, it does not have the same potential to accumulate as bisphenyl A.[428] Triclosan has been identified as an endocrine disruptor, therefore, it has the potential to affect the function of numerous organs, such as the thyroid and reproductive organs. It has been associated with dysfunction of the following endocrine organs:
Thyroid
Human and animal studies have found evidence that exposure to triclosan leads to decreased levels of total triiodothyonine (T3) and thyroxine (T4).[429-434] T3 and T4 are the thyroid hormones used to stimulate the thyroid receptors of all the cells of the body to drive all metabolic processes. Thyroid hormones are critical for normal growth and development of humans and animals; the developing brain of a child is particularly vulnerable to damage caused by disruption of the thyroid system. Studies have shown abnormal development of offspring of animals exposed to triclosan.[435-439]
 Endocrine disruption and breast cancer
Endocrine disruption and breast cancer
Triclosan may also disrupt other critical hormone systems. Recent evidence from various animal studies suggests that triclosan may have previously unrecognized endocrine disrupting properties. Research has predominantly suggested impacts to the reproductive system.[440-446] A 2008 lab study found triclosan to exert both estrogenic and androgenic effects on human breast cancer cells. [447]
 Immunosuppressive Effects
Immunosuppressive Effects
Human natural killer (NK) cells are a first line immune defense against tumor cells and virally infected cells. If their function is impaired, it leaves an individual more susceptible to cancer development or viral infection. The ability of environmental compounds, such as triclosan, to suppress the function of NK cells could contribute to increased risk of cancer development. A 2011 lab study found that triclosan diminished the function of human NK cells’ ability to lyse (kill) cancer cells by 87%![448]
A recent review reported that studies support an increase in peanut allergy prevalence in children and exposure to triclosan and having certain genetic mutations associated with food sensitization![449]
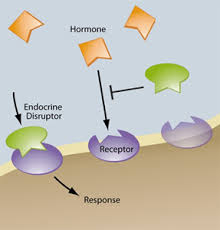 Cancer
Cancer
Recent evidence suggests that triclosan exposure may alter cancer risk, although human studies are lacking in both number and scope.[450] Three studies have investigated triclosan’s estrogenic action in cultured cancer cells suggesting that triclosan may induce proliferation but also inhibit cell proliferation in the presence of estradiol.[451-453] One of these studies added confirmation that triclosan is intrinsically estrogenic at concentrations consistent with those detected in humans.[454] Triclosan’s actions are similar to other xenoestrogens such as bisphenol A, parabens, and polychlorinated biphenyls in that it is able to bind to estrogen receptors and induce proliferation in cultured estrogen-sensitive breast cancer cells.[455,456] However, whether triclosan raises or lowers risk of cancer through estrogen-related pathways, and possible effect modification by estradiol, have not been examined in human studies.
Animal studies of triclosan exposure and cancer development have included long-term studies of its carcinogenicity but observed no increase in carcinogenesis.[457] However, one study did observe the appearance of liver tumors in mice following 18 m onths of exposure to 0–200 mg/kg/day triclosan in diet.[458] Overall, however, the results of animal studies to date have been mixed, showing null, inverse, and positive associations.[459]
onths of exposure to 0–200 mg/kg/day triclosan in diet.[458] Overall, however, the results of animal studies to date have been mixed, showing null, inverse, and positive associations.[459]
Recent research shows that triclosan in tap water can react with residual chlorine from standard water disinfecting procedures to form a variety of chlorinated byproducts at low levels, including chloroform, a suspected human carcinogen.[460] A study published in 2007 illustrated that, under some circumstances, triclosan triggered the production of chloroform in amounts up to 40% higher than background levels in chlorine-treated tap water.[461] Triclosan has also been shown to degrade into a form of dioxin, a class of chemicals linked to a broad range of toxicities including cancer.[462]
How is Triclosan regulated?
Triclosan is regulated by FDA when used in cosmetics and for medical applications, by the U.S. Environmental Protection Agency (EPA) when used as an antibacterial pesticide (in products that make “antibacterial” claims), and by the Consumer Product Safety Commission (CPSC) when used in ordinary consumer products that do not make specific “antibacterial” claims. Current regulations do not take into account concerns regarding hormone disruption or formation of toxic byproducts, nor do they fully account for the potential for multiple exposures through the broad variety of triclosan-treated everyday products.[463]
 How to Avoid Exposure: Switch to triclosan-free soaps, cosmetics and body care products. Avoid body care products, especially liquid hand soaps, labeled “antibacterial,” and always check the list of ingredients. If you use a skin disinfectant, use an alcohol hand rub or rinse. Avoid “antibacterial” dishwashing detergents, cleaning products, and other items – there is no evidence that these products protect your health. Triclosan is approved for use in over 140 different types of products, and most of these are not required to have ingredient labels. Vague claims such as “protection against mold,” “odor-fighting” or “keeps food fresher, longer,” may also indicate the presence of triclosan. Because triclosan is found in many everyday products, some exposure may be unavoidable. [464]
How to Avoid Exposure: Switch to triclosan-free soaps, cosmetics and body care products. Avoid body care products, especially liquid hand soaps, labeled “antibacterial,” and always check the list of ingredients. If you use a skin disinfectant, use an alcohol hand rub or rinse. Avoid “antibacterial” dishwashing detergents, cleaning products, and other items – there is no evidence that these products protect your health. Triclosan is approved for use in over 140 different types of products, and most of these are not required to have ingredient labels. Vague claims such as “protection against mold,” “odor-fighting” or “keeps food fresher, longer,” may also indicate the presence of triclosan. Because triclosan is found in many everyday products, some exposure may be unavoidable. [464]
References
1. http://www.ewg.org/research/dirty-dozen-list-endocrine-disruptors
2. Ehrlich S, Calafat AM, Humblet O, Smith T, Hauser R. Handling of thermal receipts as a source of exposure to bisphenol A. JAMA. 2014 Feb 26;311(8):859-60. doi: 10.1001/jama.2013.283735.
3. http://www.niehs.nih.gov/health/assets/docs_a_e/bisphenol-a-factsheet.pdf
4. Laura N. Vandenberg, Ibrahim Chahoud, Jerrold J. Heindel, Vasantha Padmanabhan, Francisco J.R. Paumgartten, Gilbert Schoenfelder. Urinary, Circulating, and Tissue Biomonitoring Studies Indicate Widespread Exposure to Bisphenol A. Environ Health Perspect. 2010 August; 118(8): 1055–1070. Published online 2010 March 24. doi: 10.1289/ehp.0901716
5. http://www.ewg.org/research/timeline-bpa-invention-phase-out
6. http://www.ewg.org/research/timeline-bpa-invention-phase-out
7. Rochester JR. Bisphenol A and human health: a review of the literature. Reprod Toxicol. 2013 Dec;42:132-55. doi: 10.1016/j.reprotox.2013.08.008. Epub 2013 Aug 30.
8. Lazúrová Z, Lazúrová I. [The environmental estrogen bisphenol A and its effects on the human organism]. Vnitr Lek. 2013 Jun;59(6):466-71. Czech.
9. Kang HJ, Hong YB, Yi YW, Cho CH, Wang A, Bae I. Correlations between BRCA1 defect and environmental factors in the risk of breast cancer. J Toxicol Sci. 2013;38(3):355-61.
10. Fernandez SV, Huang Y, Snider KE, Zhou Y, Pogash TJ, Russo J. Expression and DNA methylation changes in human breast epithelial cells after bisphenol A exposure. Int J Oncol. 2012 Jul;41(1):369-77. doi: 10.3892/ijo.2012. 1444. Epub 2012 Apr 20.
11. Sprague BL, Trentham-Dietz A, Hedman CJ, Wang J, Hemming JD, Hampton JM, Buist DS, Aiello Bowles EJ, Sisney GS, Burnside ES. Circulating serum xenoestrogens and mammographic breast density. Breast Cancer Res. 2013 May 27;15(3):R45. [Epub ahead of print]
12. Kristen Weber Lozada, Ruth A. Keri. Bisphenol A Increases Mammary Cancer Risk in Two Distinct Mouse Models of Breast Cancer. Biol Reprod. 2011 September; 85(3): 490–497. Published online 2011 June 2. doi: 10.1095/biolreprod.110.090431
13. Bhan A, Hussain I, Ansari KI, Bobzean SA, Perrotti LI, Mandal SS. Bisphenol-A and diethylstilbestrol exposure induces the expression of breast cancer associated long noncoding RNA HOTAIR in vitro and in vivo. J Steroid Biochem Mol Biol. 2014 Feb 14. pii: S0960-0760(14)00031-4. doi:10.1016/j.jsbmb.2014.02.002. [Epub ahead of print] Carcinogenesis. 2011 Nov;32(11):1724-33. doi: 10.1093/carcin/bgr196. Epub 2011 Sep 1.
14. Soto AM, Sonnenschein C. Environmental causes of cancer: endocrine disruptors as carcinogens. Nat Rev Endocrinol. 2010 Jul;6(7):363-70. doi: 10.1038/nrendo.2010.87. Epub 2010 May 25. Review.
15. Tarapore P, Ying J, Ouyang B, Burke B, Bracken B, Ho SM. Exposure to bisphenol a correlates with early-onset prostate cancer and promotes centrosome amplification and anchorage-independent growth in vitro. PLoS One. 2014 Mar 3;9(3):e90332. doi: 10.1371/journal.pone.0090332. eCollection 2014.
16. Prins GS, Hu WY, Shi GB, Hu DP, Majumdar S, Li G, Huang K, Nelles JL, Ho SM, Walker CL, Kajdacsy-Balla A, van Breemen RB. Bisphenol A promotes human prostate stem-progenitor cell self-renewal and increases in vivo carcinogenesis in human prostate epithelium. Endocrinology. 2014 Mar;155(3):805-17. doi: 10.1210/en.2013-1955. Epub 2014 Jan 1.
17. Prins GS, Birch L, Tang WY, Ho SM. Developmental estrogen exposures predispose to prostate carcinogenesis with aging. Reprod Toxicol. 2007;23:374–382.
18. Bosland MC, et al. Multistage prostate carcinogenesis: the role of hormones. Princess Takamatsu Symp. 1991;22:109–123.
19. Skakkebaek NE, Rajpert-De Meyts E, Main KM. Testicular dysgenesis syndrome: an increasingly common developmental disorder with environmental aspects. Hum Reprod. 2001;16:972–978.
20. Hardell L, Bavel B, Lindstrom G, Eriksson M, Carlberg M. In utero exposure to persistent organic pollutants in relation to testicular cancer risk. Int J Androl. 2006;29:228–234.
21. Rezg R, El-Fazaa S, Gharbi N, Mornagui B. Bisphenol A and human chronic diseases: Current evidences, possible mechanisms, and future perspectives. Environ Int. 2014 Mar;64C:83-90. doi: 10.1016/j.envint.2013.12.007. Epub 2013 Dec 29. Review.
22. Valvi D, Casas M, Mendez MA, Ballesteros-Gómez A, Luque N, Rubio S, Sunyer J, Vrijheid M. Prenatal bisphenol a urine concentrations and early rapid growth and overweight risk in the offspring. Epidemiology. 2013 Nov;24(6):791-9. doi: 10.1097/EDE.0b013e3182a67822.
23. Eng DS, Lee JM, Gebremariam A, Meeker JD, Peterson K, Padmanabhan V. Bisphenol A and chronic disease risk factors in US children. Pediatrics. 2013 Sep;132(3):e637-45. doi: 10.1542/peds.2013-0106. Epub 2013 Aug 19.
24. Lazúrová Z, Lazúrová I. [The environmental estrogen bisphenol A and its effects on the human organism]. Vnitr Lek. 2013 Jun;59(6):466-71. Czech.
25. Bhandari R, Xiao J, Shankar A. Urinary bisphenol A and obesity in U.S. children. Am J Epidemiol. 2013 Jun 1;177(11):1263-70. doi: 10.1093/aje/kws391. Epub 2013 Apr 4.
26. Fenichel P, Chevalier N, Brucker-Davis F. Bisphenol A: an endocrine and metabolic disruptor. Ann Endocrinol (Paris). 2013 Jul;74(3):211-20. doi: 10.1016/j.ando.2013.04.002. Epub 2013 Jun 21.
27. Chen M, Zhou K, Chen X, Qiao S, Hu Y, Xu B, Xu B, Han X, Tang R, Mao Z, Dong C, Wu D, Wang Y, Wang S, Zhou Z, Xia Y, Wang X. Metabolomic Analysis Reveals Metabolic Changes Caused by Bisphenol A in Rats. Toxicol Sci. 2014 Feb 20. [Epub ahead of print]
28. Frederick S. vom Saal, Susan C. Nagel, Benjamin L. Coe, Brittany M. Angle, Julia A. Taylor. The Estrogenic Endocrine Disrupting Chemical Bisphenol A (BPA) And Obesity. Mol Cell Endocrinol. Author manuscript; available in PMC 2013 May 6. Published in final edited form as: Mol Cell Endocrinol. 2012 May 6; 354(1-2): 74–84.
29. Monica K. Silver, Marie S. O’Neill, MaryFran R. Sowers, Sung Kyun Park. Urinary Bisphenol A and Type-2 Diabetes in U.S. Adults: Data from NHANES 2003-2008. PLoS One. 2011; 6(10): e26868. Published online 2011 October 26. doi: 10.1371/journal.pone.0026868
30. Khalil N, Ebert JR, Wang L, Belcher S, Lee M, Czerwinski SA, Kannan K. Bisphenol A and cardiometabolic risk factors in obese children. Sci Total Environ. 2014 Feb 1;470-471:726-32. doi: 10.1016/j.scitotenv.2013.09.088. Epub 2013 Oct 30.
31. Kim MJ, Moon MK, Kang GH, Lee KJ, Choi SH, Lim S, Oh BC, Park DJ, Park KS, Jang HC, Park YJ. Chronic Exposure to Bisphenol A can Accelerate Atherosclerosis in High-Fat-Fed Apolipoprotein E Knockout Mice. Cardiovasc Toxicol. 2013 Nov 14. [Epub ahead of print]
32. Zeliger HI. Lipophilic chemical exposure as a cause of cardiovascular disease. Interdiscip Toxicol. 2013 Jun;6(2):55-62. Review.
33. Melzer D, Rice NE, Lewis C, Henley WE, Galloway TS. Association of urinary bisphenol A concentration with heart disease: evidence from NHANES 2003/06. PLoS One. 2010 Jan 13;5(1):e8673. doi: 10.1371/journal.pone.0008673.
34. Melzer D, Osborne NJ, Henley WE, Cipelli R, Young A, Money C, McCormack P, Luben R, Khaw KT, Wareham NJ, Galloway TS. Urinary bisphenol A concentration and risk of future coronary artery disease in apparently healthy men and women. Circulation. 2012 Mar 27;125(12):1482-90. doi: 10.1161/ CIRCULATIONAHA.111.069153. Epub 2012 Feb 21.
35. Yıldız N, Barlas N. Hepatic and renal functions in growing male rats after bisphenol A and octylphenol exposure. Hum Exp Toxicol. 2013 Jul;32(7):675-86. doi: 10.1177/0960327112464796.
36. Jayashree S, Indumathi D, Akilavalli N, Sathish S, Selvaraj J, Balasubramanian K. Effect of Bisphenol-A on insulin signal transduction and glucose oxidation in liver of adult male albino rat. Environ Toxicol Pharmacol. 2013 Mar;35(2):300-10. doi: 10.1016/j.etap.2012.12.016. Epub 2013 Jan 11.
37. Hassan ZK, Elobeid MA, Virk P, Omer SA, ElAmin M, Daghestani MH, AlOlayan EM. Bisphenol A induces hepatotoxicity through oxidative stress in rat model. Oxid Med Cell Longev. 2012;2012:194829. doi: 10.1155/2012/194829. Epub 2012 Jul 24.
38. Moon MK, Kim MJ, Jung IK, Koo YD, Ann HY, Lee KJ, Kim SH, Yoon YC, Cho BJ, Park KS, Jang HC, Park YJ. Bisphenol A impairs mitochondrial function in the liver at doses below the no observed adverse effect level. J Korean Med Sci. 2012 Jun;27(6):644-52. doi: 10.3346/jkms.2012.27.6.644. Epub 2012 May 26.
39. Castro B, Sánchez P, Torres JM, Preda O, del Moral RG, Ortega E. Bisphenol A exposure during adulthood alters expression of aromatase and 5α-reductase isozymes in rat prostate. PLoS One. 2013;8(2):e55905. doi: 10.1371/journal.pone.0055905. Epub 2013 Feb 6.
40. Timms BG, et al. Estrogenic chemicals in plastic and oral contraceptives disrupt development of the fetal mouse prostate and urethra. Proc Natl Acad Sci USA. 2005;102:7014–7019.
41. Timms BG, Peterson RE, vom Saal FS. 2,3,7,8-Tetrachlorodibenzo-p-dioxin interacts with endogenous estradiol to disrupt prostate gland morphogenesis in male rat fetuses. Toxicol Sci. 2002;67:264–274.
42. Huang L, Pu Y, Alam S, Birch L, Prins GS. Estrogenic regulation of signaling pathways and homeobox genes during rat prostate development. J Androl. 2004;25:330–337.
43. Dobrzyńska MM, Jankowska-Steifer EA, Tyrkiel EJ, Gajowik A, Radzikowska J, Pachocki KA. Comparison of the effects of bisphenol A alone and in a combination with X-irradiation on sperm count and quality in male adult and pubescent mice. Environ Toxicol. 2013 Apr 25. doi: 10.1002/tox.21861. [Epub ahead of print]
44. Qiu LL, Wang X, Zhang XH, Zhang Z, Gu J, Liu L, Wang Y, Wang X, Wang SL. Decreased androgen receptor expression may contribute to spermatogenesis failure in rats exposed to low concentration of bisphenol A. Toxicol Lett. 2013 May 23;219(2):116-24. doi: 10.1016/j.toxlet.2013.03.011. Epub 2013 Mar 22.
45. Meeker JD, Ehrlich S, Toth TL, Wright DL, Calafat AM, Trisini AT, Ye X, Hauser R. Semen quality and sperm DNA damage in relation to urinary bisphenol A among men from an infertility clinic. Reprod Toxicol. 2010 Dec;30(4):532-9. doi: 10.1016/j.reprotox.2010.07.005. Epub 2010 Jul 23.
46. Rutkowska A, Rachoń D. Bisphenol A (BPA) and its potential role in the pathogenesis of the polycystic ovary syndrome (PCOS). Gynecol Endocrinol. 2014 Jan 7. [Epub ahead of print]
47. Richter CA, Birnbaum LS, Farabollini F, Newbold RR, Rubin BS, Talsness CE, Vandenbergh JG, Walser-Kuntz DR, Vom Saal FS. In vivo effects of bisphenol A in laboratory rodent studies. Reprod Toxicol 2007; 24: 199–224..224.
48. Vandenberg LN, Maffini MV, Sonnenschein C, Rubin BS, Soto AM. Bisphenol-A and the great divide: a review of controversies in the field of endocrine disruption. Endocr Rev 2009; 30: 75–95..95.
49. Susiarjo M, Hassold TJ, Freeman E, Hunt PA. Bisphenol A exposure in utero disrupts early oogenesis in the mouse. PLoS Genet 2007; 3: e5.
50. Vandenberg LN, Maffini MV, Schaeberle CM, Ucci AA, Sonnenschein C, Rubin BS, Soto AM. Perinatal exposure to the xenoestrogen bisphenol-A induces mammary intraductal hyperplasias in adult CD-1 mice. Reprod Toxicol 2008; 26: 210–219..219.
51. Newbold RR, Jefferson WN, Padilla-Banks E. Long-term adverse effects of neonatal exposure to bisphenol A on the murine female reproductive tract. Reprod Toxicol. 2007 Aug-Sep;24(2):253-8. Epub 2007 Jul 27.
52. Lawson C, Gieske M, Murdoch B, Ye P, Li Y, Hassold T, Hunt PA. Gene expression in the fetal mouse ovary is altered by exposure to low doses of bisphenol A. Biol Reprod. 2011 Jan;84(1):79-86. doi:10.1095/biolreprod.110.084814. Epub 2010 Aug 25.
53. Patricia A. Hunt, Martha Susiarjo, Carmen Rubio, Terry J. Hassold. The Bisphenol A Experience: A Primer for the Analysis of Environmental Effects on Mammalian Reproduction. Biol Reprod. 2009 November; 81(5): 807–813. Published online 2009 May 20. doi: 10.1095/biolreprod.109.077008
54. Alonso-Magdalena P, Vieira E, Soriano S, Menes L, Burks D, Quesada I, Nadal A. Bisphenol A exposure during pregnancy disrupts glucose homeostasis in mothers and adult male offspring. Environ Health Perspect. 2010 Sep;118(9):1243-50. doi: 10.1289/ehp.1001993. Epub 2010 May 7.
55. Markey CM, Rubin BS, Soto AM, Sonnenschein C. Endocrine disruptors: from Wingspread to environmental developmental biology. J Steroid Biochem Mol Biol. 2002 Dec;83(1-5):235-44. Review.
56. Susiarjo M, Hassold TJ, Freeman E, Hunt PA. Bisphenol A exposure in utero disrupts early oogenesis in the mouse. PLoS Genet. 2007 Jan 12;3(1):e5.
57. Richter CA, Taylor JA, Ruhlen RL, Welshons WV, Vom Saal FS. Estradiol and Bisphenol A stimulate androgen receptor and estrogen receptor gene expression in fetal mouse prostate mesenchyme cells. Environ Health Perspect. 2007 Jun;115(6):902-8. Epub 2007 Feb 27.
58. Komada M, Asai Y, Morii M, Matsuki M, Sato M, Nagao T. Maternal bisphenol A oral dosing relates to the acceleration of neurogenesis in the developing neocortex of mouse fetuses. Toxicology. 2012 May 16;295(1-3):31-8. doi: 10.1016/j.tox.2012.02.013. Epub 2012 Mar 7.
59. Braun JM, Hauser R. Bisphenol A and children’s health. Curr Opin Pediatr. 2011 Apr;23(2):233-9. doi: 10.1097/MOP.0b013e3283445675. Review.
60. http://www.ewg.org/research/tips-avoid-bpa-exposure
61. Kajta M, Wójtowicz AK. Impact of endocrine-disrupting chemicals on neural development and the onset of neurological disorders. Pharmacol Rep. 2013;65(6):1632-9.
62. León-Olea M, Martyniuk CJ, Orlando EF, Ottinger MA, Rosenfeld C, Wolstenholme J, Trudeau VL. Current concepts in neuroendocrine disruption. Gen Comp Endocrinol. 2014 Feb 13. pii: S0016-6480(14)00048-3. doi: 10.1016/j.ygcen.2014.02.005. [Epub ahead of print]
63. Elsworth JD, Jentsch JD, Vandevoort CA, Roth RH, Jr DE, Leranth C. Prenatal exposure to bisphenol A impacts midbrain dopamine neurons and hippocampal spine synapses in non-human primates. Neurotoxicology. 2013 Mar;35:113-20. doi: 10.1016/j.neuro.2013.01.001. Epub 2013 Jan 18.
64. Viberg H, Lee I. A single exposure to bisphenol A alters the levels of important neuroproteins in adult male and female mice. Neurotoxicology. 2012 Oct;33(5):1390-5. doi: 10.1016/j.neuro.2012.09.002. Epub 2012 Sep 12.
65. Weiss B. The intersection of neurotoxicology and endocrine disruption. Neurotoxicology. 2012 Dec;33(6):1410-9. doi: 10.1016/j.neuro.2012.05.014. Epub 2012 May 31. Review.
66. Jang YJ, Park HR, Kim TH, Yang WJ, Lee JJ, Choi SY, Oh SB, Lee E, Park JH, Kim HP, Kim HS, Lee J. High dose bisphenol A impairs hippocampal neurogenesis in female mice across generations. Toxicology. 2012 Jun 14;296(1-3):73-82. doi: 10.1016/j.tox.2012.03.007. Epub 2012 Apr 3.
67. Komada M, Asai Y, Morii M, Matsuki M, Sato M, Nagao T. Maternal bisphenol A oral dosing relates to the acceleration of neurogenesis in the developing neocortex of mouse fetuses. Toxicology. 2012 May 16;295(1-3):31-8. doi: 10.1016/j.tox.2012.02.013. Epub 2012 Mar 7.
68. Itoh K, Yaoi T, Fushiki S. Bisphenol A, an endocrine-disrupting chemical, and brain development. Neuropathology. 2012 Aug;32(4):447-57. doi: 10.1111/j.1440-1789.2011.01287.x. Epub 2012 Jan 12. Review.
69. Longnecker MP. Human data on bisphenol A and neurodevelopment. Environ Health Perspect. 2009 Dec;117(12):A531-2. doi: 10.1289/ehp.0901610. No abstract available.
70. Leranth C, Hajszan T, Szigeti-Buck K, Bober J, MacLusky NJ. Bisphenol A prevents the synaptogenic response to estradiol in hippocampus and prefrontal cortex of ovariectomized nonhuman primates. Proc Natl Acad Sci U S A. 2008 Sep 16;105(37):14187-91. doi: 10.1073/pnas.0806139105. Epub 2008 Sep 3.
71. Kundakovic M, Gudsnuk K, Franks B, Madrid J, Miller RL, Perera FP, Champagne FA. Sex-specific epigenetic disruption and behavioral changes following low-dose in utero bisphenol A exposure. Proc Natl Acad Sci U S A. 2013 Jun 11;110(24):9956-61. doi: 10.1073/pnas.1214056110. Epub 2013 May 28.
72. Hong SB, Hong YC, Kim JW, Park EJ, Shin MS, Kim BN, Yoo HJ, Cho IH, Bhang SY, Cho SC. Bisphenol A in relation to behavior and learning of school-age children. J Child Psychol Psychiatry. 2013 Aug;54(8):890-9. doi: 10.1111/jcpp.12050. Epub 2013 Feb 28.
73. Xu X, Hong X, Xie L, Li T, Yang Y, Zhang Q, Zhang G, Liu X. Gestational and lactational exposure to bisphenol-A affects anxiety- and depression-like behaviors in mice. Horm Behav. 2012 Sep;62(4):480-90.
74. Wolstenholme JT, Edwards M, Shetty SR, Gatewood JD, Taylor JA, Rissman EF, Connelly JJ. Gestational exposure to bisphenol A produces transgenerational changes in behaviors and gene expression. Endocrinology. 2012 Aug;153(8):3828-38. doi: 10.1210/en.2012-1195. Epub 2012 Jun 15.
75. Fudvoye J, Bourguignon JP, Parent AS. Endocrine-disrupting chemicals and human growth and maturation: a focus on early critical windows of exposure. Vitam Horm. 2014;94:1-25. doi: 10.1016/B978-0-12-800095-3.00001-8.
76. Beverly S. Rubin, Ana M. Soto. Bisphenol A: Perinatal exposure and body weight. Mol Cell Endocrinol. Author manuscript; available in PMC 2010 February 9.
77. vom Saal FS, Hughes C. An extensive new literature concerning low-dose effects of bisphenol A shows the need for a new risk assessment. Environ Health Perspect 2005; 113: 926–933..933.
78. http://www.ewg.org/research/tips-avoid-bpa-exposure
79. http://www.ewg.org/research/tips-avoid-bpa-exposure
80. http://www.ewg.org/dioxin/analysis
81. http://www.who.int/mediacentre/factsheets/fs225/en/
82. http://www.who.int/mediacentre/factsheets/fs225/en/
83. http://www.who.int/mediacentre/factsheets/fs225/en/
85. Marinković N, Pašalić D, Ferenčak G, Gršković B, Stavljenić Rukavina A. Dioxins and human toxicity. Arh Hig Rada Toksikol. 2010 Dec;61(4):445-53. doi: 10.2478/10004-1254-61-2010-2024. Review.
86. Bethany Winans, Michael C. Humble, B. Paige Lawrence. Environmental toxicants and the developing immune system: a missing link in the global battle against infectious disease? Reprod Toxicol. Author manuscript; available in PMC 2012 April 1. Published in final edited form as: Reprod Toxicol. 2011 April; 31(3): 327–336. Published online 2010 September 22. doi: 10.1016/j.reprotox.2010.09.004
87. Nagayama J, Tsuji H, Iida T, Nakagawa R, Matsueda T, Hirakawa H, et al. Immunologic effects of perinatal exposure to dioxins, PCBs and organochlorine pesticides in Japanese infants. Chemosphere. 2007;67:S393–8.
88. ten Tusscher GW, Steerenberg PA, van Loveren H, Vos JG, Borne AE, Westra M, et al. Persistent hematologic and immunologic disturbances in 8-year-old Dutch children associated with perinatal dioxin exposure. Environ Health Perspect. 2003;111:1519–23.
89. Weisglas-Kuperus N, Sas TC, Koopman-Esseboom C, Zwan CW, De Ridder MA, Beishuizen A, et al. Immunologic effects of background prenatal and postnatal exposure to dioxins and polychlorinated biphenyls in Dutch infants. Pediatr Res. 1995;38:404–10.
90. Weisglas-Kuperus N, Patandin S, Berbers GA, Sas TC, Mulder PG, Sauer PJ, et al. Immunologic effects of background exposure to polychlorinated biphenyls and dioxins in Dutch preschool children. Environmental health perspectives. 2000;108:1203.
91. Weisglas-Kuperus N, Vreugdenhil HJ, Mulder PG. Immunological effects of environmental exposure to polychlorinated biphenyls and dioxins in Dutch school children. Toxicol Lett. 2004;149:281–5.
92. Hertz-Picciotto I, Park HY, Dostal M, Kocan A, Trnovec T, Sram R. Prenatal exposures to persistent and non-persistent organic compounds and effects on immune system development. Basic & clinical pharmacology & toxicology. 2008;102:146–54.
93. Gascon M, Morales E, Sunyer J, Vrijheid M. Effects of persistent organic pollutants on the developing respiratory and immune systems: a systematic review. Environ Int. 2013 Feb;52:51-65. doi: 10.1016/j.envint.2012.11.005. Epub 2013 Jan 3. Review.
94. Carpenter DO, Ma J, Lessner L. Asthma and infectious respiratory disease in relation to residence near hazardous waste sites. Ann N Y Acad Sci. 2008 Oct;1140:201-8. doi: 10.1196/annals.1454.000.
95. Kasai T, Saito M, Senoh H, Umeda Y, Aiso S, Ohbayashi H, Nishizawa T, Nagano K, Fukushima S. Thirteen-week inhalation toxicity of 1,4-dioxane in rats. Inhal Toxicol. 2008 Aug;20(10):961-71. doi: 10.1080/08958370802105397 .
96. Wong PS, Vogel CF, Kokosinski K, Matsumura F. Arylhydrocarbon receptor activation in NCI-H441 cells and C57BL/6 mice: possible mechanisms for lung dysfunction. Am J Respir Cell Mol Biol. 2010 Feb;42(2):210-7. doi: 10.1165/rcmb.2008-0228OC. Epub 2009 Apr 16.
97. Kransler KM, McGarrigle BP, Swartz DD, Olson JR. Lung development in the Holtzman rat is adversely affected by gestational exposure to 2,3,7,8-tetrachlorodibenzo-p-dioxin. Toxicol Sci. 2009 Feb;107(2):498-511. doi: 10.1093/toxsci/kfn235. Epub 2008 Nov 6.
98. Walker NJ, Yoshizawa K, Miller RA, Brix AE, Sells DM, Jokinen MP, Wyde ME, Easterling M, Nyska A. Pulmonary lesions in female Harlan Sprague-Dawley rats following two-year oral treatment with dioxin-like compounds. Toxicol Pathol. 2007 Dec;35(7):880-9.
99. ten Tusscher GW, Koppe JG. Perinatal dioxin exposure and later effects–a review. Chemosphere. 2004 Mar;54(9):1329-36. Review.
100. Fernández-González R, Yebra-Pimentel I, Martínez-Carballo E, Simal-Gándara J. A critical review about the human exposure to polychlorinated dibenzo-p-dioxins (PCDDs), polychlorinated dibenzofurans (PCDFs) and polychlorinated biphenyls (PCBs) through foods. Crit Rev Food Sci Nutr. 2013 Nov 26. [Epub ahead of print]
101. Xu HM, Xie HQ, Tao WQ, Zhou ZG, Li SZ, Zhao B. Dioxin and Dioxin-Like Compounds Suppress Acetylcholinesterase Activity via Transcriptional Downregulations In Vitro. J Mol Neurosci. 2013 Nov 17. [Epub ahead of print]
102. Takeda T, Fujii M, Hattori Y, Yamamoto M, Shimazoe T, Ishii Y, Himeno M, Yamada H. Maternal exposure to dioxin imprints sexual immaturity of the pups through fixing the status of the reduced expression of hypothalamic gonadotropin-releasing hormone. Mol Pharmacol. 2014 Jan;85(1):74-82. doi: 10.1124/mol.113.088575. Epub 2013 Oct 16.
103. Nguyen MN, Nishijo M, Nguyen AT, Bor A, Nakamura T, Hori E, Nakagawa H, Ono T, Nishijo H. Effects of maternal exposure to 2,3,7,8-tetrachlorodibenzo-p-dioxin on parvalbumin- and calbindin-immunoreactive neurons in the limbic system and superior colliculus in rat offspring. Toxicology. 2013 Dec 6;314(1):125-34. doi: 10.1016/j.tox.2013.09.005. Epub 2013 Sep 20.
104. Xie HQ, Xu HM, Fu HL, Hu Q, Tian WJ, Pei XH, Zhao B. AhR-mediated effects of dioxin on neuronal acetylcholinesterase expression in vitro. Environ Health Perspect. 2013 May;121(5):613-8. doi: 10.1289/ehp.1206066. Epub 2013 Feb 19.
105. Iida M, Kim EY, Murakami Y, Shima Y, Iwata H. Toxic effects of 2,3,7,8-tetrachlorodibenzo-p-dioxin on the peripheral nervous system of developing red seabream (Pagrus major). Aquat Toxicol. 2013 Mar 15;128-129:193-202. doi: 10.1016/j.aquatox.2012.12.009. Epub 2012 Dec 21.
106. Endo T, Kakeyama M, Uemura Y, Haijima A, Okuno H, Bito H, Tohyama C. Executive function deficits and social-behavioral abnormality in mice exposed to a low dose of dioxin in utero and via lactation. PLoS One. 2012;7(12):e50741. doi: 10.1371/journal.pone.0050741. Epub 2012 Dec 12.
107. Tomasini MC, Beggiato S, Ferraro L, Tanganelli S, Marani L, Lorenzini L, Antonelli T. Prenatal exposure to 2,3,7,8-tetrachlorodibenzo-p-dioxin produces alterations in cortical neuron development and a long-term dysfunction of glutamate transmission in rat cerebral cortex. Neurochem Int. 2012 Oct;61(5):759-66. doi: 10.1016/j.neuint.2012.07.004. Epub 2012 Jul 10.
108. Fernández-González R, Yebra-Pimentel I, Martínez-Carballo E, Simal-Gándara J. A critical review about the human exposure to polychlorinated dibenzo-p-dioxins (PCDDs), polychlorinated dibenzofurans (PCDFs) and polychlorinated biphenyls (PCBs) through foods. Crit Rev Food Sci Nutr. 2013 Nov 26. [Epub ahead of print]
109. Wong MH, Armour MA, Naidu R, Man M. Persistent toxic substances: sources, fates and effects. Rev Environ Health. 2012;27(4):207-13. doi: 10.1515/reveh-2012-0040. Review.
110. Rysavy NM, Maaetoft-Udsen K, Turner H. Dioxins: diagnostic and prognostic challenges arising from complex mechanisms. J Appl Toxicol. 2013 Jan;33(1):1-8. doi: 10.1002/jat.2759. Epub 2012 May 19. Review.
111. Schug TT, Janesick A, Blumberg B, Heindel JJ. Endocrine disrupting chemicals and disease susceptibility. J Steroid Biochem Mol Biol. 2011 Nov;127(3-5):204-15. doi: 10.1016/j.jsbmb.2011.08.007. Epub 2011 Aug 27. Review.
112. Trnovec T, Jusko TA, Šovčíková E, Lancz K, Chovancová J, Patayová H, Palkovičová L, Drobná B, Langer P, Van den Berg M, Dedik L, Wimmerová S. Relative effect potency estimates of dioxin-like activity for dioxins, furans, and dioxin-like PCBs in adults based on two thyroid outcomes. Environ Health Perspect. 2013 Aug;121(8):886-92. doi: 10.1289/ehp.1205739. Epub 2013 May 1.
113. Rennert A, Wittsiepe J, Kasper-Sonnenberg M, Binder G, Fürst P, Cramer C, Krämer U, Wilhelm M. Prenatal and early life exposure to polychlorinated dibenzo-p-dioxins, dibenzofurans and biphenyls may influence dehydroepiandrosterone sulfate levels at prepubertal age: results from the Duisburg birth cohort study. J Toxicol Environ Health A. 2012;75(19-20):1232-40.
114. Craig ZR, Wang W, Flaws JA. Endocrine-disrupting chemicals in ovarian function: effects on steroidogenesis, metabolism and nuclear receptor signaling. Reproduction. 2011 Nov;142(5):633-46. doi: 10.1530/REP-11-0136. Epub 2011 Aug 23. Review.
115. Balabanič D, Rupnik M, Klemenčič AK. Negative impact of endocrine-disrupting compounds on human reproductive health. Reprod Fertil Dev. 2011;23(3):403-16. doi: 10.1071/RD09300. Review.
116. Fernández-González R, Yebra-Pimentel I, Martínez-Carballo E, Simal-Gándara J. A critical review about the human exposure to polychlorinated dibenzo-p-dioxins (PCDDs), polychlorinated dibenzofurans (PCDFs) and polychlorinated biphenyls (PCBs) through foods. Crit Rev Food Sci Nutr. 2013 Nov 26. [Epub ahead of print]
117. Balabanič D, Rupnik M, Klemenčič AK. Negative impact of endocrine-disrupting compounds on human reproductive health. Reprod Fertil Dev. 2011;23(3):403-16. doi: 10.1071/RD09300. Review.
118. Papadopoulou E, Vafeiadi M, Agramunt S, Mathianaki K, Karakosta P, Spanaki A, Besselink H, Kiviranta H, Rantakokko P, KaterinaSarri, Koutis A, Chatzi L, Kogevinas M. Maternal diet, prenatal exposure to dioxins and other persistent organic pollutants and anogenital distance in children. Sci Total Environ. 2013 Sep 1;461-462:222-9. doi: 10.1016/j.scitotenv.2013.05.005. Epub 2013 May 31.
119. Zawatski W, Lee MM. Male pubertal development: are endocrine-disrupting compounds shifting the norms? J Endocrinol. 2013 Jul 11;218(2):R1-12. doi: 10.1530/JOE-12-0449. Print 2013. Review.
120. Takeda T, Fujii M, Taura J, Ishii Y, Yamada H. Dioxin silences gonadotropin expression in perinatal pups by inducing histone deacetylases: a new insight into the mechanism for the imprinting of sexual immaturity by dioxin. J Biol Chem. 2012 May 25;287(22):18440-50. doi: 10.1074/jbc.M111.335158. Epub 2012 Apr 9.
121. Baker TR, Peterson RE, Heideman W. Using zebrafish as a model system for studying the transgenerational effects of dioxin. Toxicol Sci. 2014 Apr;138(2):403-11. doi: 10.1093/toxsci/kfu006. Epub 2014 Jan 27.
122. Brunnberg S, Andersson P, Poellinger L, Hanberg A. The constitutively active Ah receptor (CA-AhR) mouse as a model for dioxin exposure – effects in reproductive organs. Chemosphere. 2011 Dec;85(11):1701-6. doi: 10.1016/j.chemosphere.2011.09.015. Epub 2011 Oct 19.
123. Naville D, Rebourcet D, Chauvin MA, Vega N, Jalabert A, Vigier M, Loizon E, Bégeot M, Le Magueresse-Battistoni B. Direct and indirect impact of 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) on adult mouse Leydig cells: an in vitro study. Toxicol Lett. 2011 Dec 15;207(3):251-7. doi: 10.1016/j.toxlet.2011.09.019. Epub 2011 Sep 29.
124. Svechnikov K, Izzo G, Landreh L, Weisser J, Söder O. Endocrine disruptors and Leydig cell function. J Biomed Biotechnol. 2010;2010. pii: 684504. doi: 10.1155/2010/684504. Epub 2010 Aug 25. Review.
125. Sankpal UT, Pius H, Khan M, Shukoor MI, Maliakal P, Lee CM, Abdelrahim M, Connelly SF, Basha R. Environmental factors in causing human cancers: emphasis on tumorigenesis. Tumour Biol. 2012 Oct;33(5):1265-74. doi: 10.1007/s13277-012-0413-4. Epub 2012 May 22. Review.
126. Rysavy NM, Maaetoft-Udsen K, Turner H. Dioxins: diagnostic and prognostic challenges arising from complex mechanisms. J Appl Toxicol. 2013 Jan;33(1):1-8. doi: 10.1002/jat.2759. Epub 2012 May 19. Review.
127. Wong MH, Armour MA, Naidu R, Man M. Persistent toxic substances: sources, fates and effects. Rev Environ Health. 2012;27(4):207-13. doi: 10.1515/reveh-2012-0040. Review.
128. Słowińska M, Koter-Michalak M, Bukowska B. [The effect of dioxins on the human organism–epidemiological studies]. Med Pr. 2011;62(6):643-52. Review. Polish.
129. Bock KW. The human Ah receptor: hints from dioxin toxicities to deregulated target genes and physiological functions. Biol Chem. 2013 Jun;394(6):729-39. doi: 10.1515/hsz-2012-0340. Review.
130. Struciński P, Piskorska-Pliszczyńska J, Góralczyk K, Warenik-Bany M, Maszewski S, Czaja K, Ludwicki JK. [Dioxins and food safety]. Rocz Panstw Zakl Hig. 2011;62(1):3-17. Review. Polish.
131. Jenkins S, Betancourt AM, Wang J, Lamartiniere CA. Endocrine-active chemicals in mammary cancer causation and prevention. J Steroid Biochem Mol Biol. 2012 Apr;129(3-5):191-200. doi:10.1016/j.jsbmb.2011.06.003. Epub 2011 Jun 23. Review.
132. Donato F, Zani C. [Chronic exposure to organochlorine compounds and health effects in adults: cancer, non-Hodgkin lymphoma. Review of literature]. Ann Ig. 2010 Jul-Aug;22(4):357-67. Review. Italian.
133. Papoutsis AJ, Selmin OI, Borg JL, Romagnolo DF. Gestational exposure to the AhR agonist 2,3,7,8-tetrachlorodibenzo-p-dioxin induces BRCA-1 promoter hypermethylation and reduces BRCA-1 expression in mammary tissue of rat offspring: Preventive effects of resveratrol. Mol Carcinog. 2013 Oct 17. doi: 10.1002/mc.22095. [Epub ahead of print]
134. Hoenemeyer LA. Urologic cancer risks for veterans exposed to Agent Orange. Urol Nurs. 2013 Mar-Apr;33(2):87-90, 99.
135. http://www.ewg.org/dioxin/press
136. http://www.who.int/mediacentre/factsheets/fs225/en/
137. http://www.ewg.org/research/mouths-babes
138. Hayes, Tyrone B.; Anderson, Lloyd L.; Beasley, Val R.; de Solla, Shane R.; Iguchi, Taisen; et al. (2011). Demasculinization and feminization of male gonads by atrazine: Consistent effects across vertebrate classes. The Journal of Steroid Biochemistry and Molecular Biology 127 (1-2): 64–73. doi:10.1016/j.jsbmb.2011.03.015
139. http://www.mprnews.org/story/2007/01/02/hayesmayo
140. Fan W, Yanase T, Morinaga H, Gondo S, Okabe T, Nomura M, Komatsu T, Morohashi K, Hayes TB, Takayanagi R, Nawata H. Atrazine-induced aromatase expression is SF-1 dependent: implications for endocrine disruption in wildlife and reproductive cancers in humans. Environ Health Perspect. 2007 May;115(5):720-7. Epub 2007 Feb 5.
141. Stanko JP, Enoch RR, Rayner JL, Davis CC, Wolf DC, Malarkey DE, Fenton SE. Effects of prenatal exposure to a low dose atrazine metabolite mixture on pubertal timing and prostate development of male Long-Evans rats. Reprod Toxicol. 2010 Dec;30(4):540-9. doi: 10.1016/j.reprotox.2010.07.006. Epub 2010 Aug 19. Erratum in: Reprod Toxicol. 2011 Jul;32(1):146-7.
142. Stoker TE, Robinette CL, Cooper RL. Maternal exposure to atrazine during lactation suppresses suckling-induced prolactin release and results in prostatitis in the adult offspring. Toxicol Sci. 1999 Nov;52(1):68-79.
143. http://www.nrdc.org/health/atrazine/
144. http://toxtown.nlm.nih.gov/text_version/chemicals.php?id=24
145. http://www.cdc.gov/biomonitoring/Phthalates_FactSheet.html
146. Blount BC, Silva MJ, Caudill SP, Needham LL, Pirkle JL, Sampson EJ, Lucier GW, Jackson RJ, Brock JW. Levels of seven urinary phthalate metabolites in a human reference population. Environ Health Perspect. 2000 Oct;108(10):979-82.
147. Kohn MC, Parham F, Masten SA, Portier CJ, Shelby MD, Brock JW, Needham LL. Human exposure estimates for phthalates. Environ Health Perspect. 2000 Oct;108(10):A440-2.
148. http://www.cdc.gov/exposurereport/pdf/FourthReport.pdf
149. Wolff MS, Teitelbaum SL, Windham G, Pinney SM, Britton JA, Chelimo C, Godbold J, Biro F, Kushi LH, Pfeiffer CM, Calafat AM. Pilot study of urinary biomarkers of phytoestrogens, phthalates, and phenols in girls. Environ Health Perspect. 2007 Jan;115(1):116-21.
150. Rudel RA1, Camann DE, Spengler JD, Korn LR, Brody JG. Phthalates, alkylphenols, pesticides, polybrominated diphenyl ethers, and other endocrine-disrupting compounds in indoor air and dust. Environ Sci Technol. 2003 Oct 15;37(20):4543-53.
151. Kolpin DW, Furlong ET, Meyer MT, Thurman EM, Zaugg SD, Barber LB, Buxton HT. Pharmaceuticals, hormones, and other organic wastewater contaminants in U.S. streams, 1999-2000: a national reconnaissance. Environ Sci Technol. 2002 Mar 15;36(6):1202-11.
152. Hauser R, Meeker JD, Singh NP, Silva MJ, Ryan L, Duty S, Calafat AM. DNA damage in human sperm is related to urinary levels of phthalate monoester and oxidative metabolites. Hum Reprod. 2007 Mar;22(3):688-95. Epub 2006 Nov 7.
153. Hauser R, Meeker JD, Duty S, Silva MJ, Calafat AM. Altered semen quality in relation to urinary concentrations of phthalate monoester and oxidative metabolites. Epidemiology. 2006 Nov;17(6):682-91.
154. Duty SM, Calafat AM, Silva MJ, Ryan L, Hauser R. Phthalate exposure and reproductive hormones in adult men. Hum Reprod. 2005 Mar;20(3):604-10. Epub 2004 Dec 9.
155. Grady R, Sathyanarayana S. An update on phthalates and male reproductive development and function. Curr Urol Rep. 2012 Aug;13(4):307-10. doi: 10.1007/s11934-012-0261-1.
156. Bustamante-Montes LP, Hernández-Valero MA, Flores-Pimentel D, García-Fábila M, Amaya-Chávez A, Barr DB, Borja-Aburto VH. Prenatal exposure to phthalates is associated with decreased anogenital distance and penile size in male newborns. J Dev Orig Health Dis. 2013 Aug;4(4). doi: 10.1017/S2040174413000172.
157. Veeramachaneni DN, Klinefelter GR. Phthalate-Induced Pathology in the Foetal Testis Involves More Than Decreased Testosterone Production. Reproduction. 2013 Nov 26. [Epub ahead of print]
158. Main KM, Mortensen GK, Kaleva MM, Boisen KA, Damgaard IN, Chellakooty M, Schmidt IM, Suomi AM, Virtanen HE, Petersen DV, Andersson AM, Toppari J, Skakkebaek NE. Human breast milk contamination with phthalates and alterations of endogenous reproductive hormones in infants three months of age. Environ Health Perspect. 2006 Feb;114(2):270-6.
159. Howdeshell KL, Rider CV, Wilson VS, Gray LE Jr. Mechanisms of action of phthalate esters, individually and in combination, to induce abnormal reproductive development in male laboratory rats. Environ Res. 2008 Oct;108(2):168-76. Review.
160. Noriega NC, Howdeshell KL, Furr J, Lambright CR, Wilson VS, Gray LE Jr. Pubertal administration of DEHP delays puberty, suppresses testosterone production, and inhibits reproductive tract development in male Sprague-Dawley and Long-Evans rats. Toxicol Sci. 2009 Sep;111(1):163-78. doi:10.1093/toxsci/kfp129. Epub 2009 Jun 15.
161. Pak VM, McCauley LA, Pinto-Martin J. Phthalate exposures and human health concerns: A review and implications for practice. AAOHN J. 2011 May;59(5):228-33; quiz 234-5. doi: 10.3928/08910162-20110426-01. Review.
162. Crinnion WJ. Toxic effects of the easily avoidable phthalates and parabens. Altern Med Rev. 2010 Sep;15(3):190-6. Review.
163. Callesen M, Bekö G, Weschler CJ, Langer S, Brive L, Clausen G, Toftum J, Sigsgaard T, Høst A, Jensen TK. Phthalate metabolites in urine and asthma, allergic rhinoconjunctivitis and atopic dermatitis in preschool children. Int J Hyg Environ Health. 2013 Dec 13. pii: S1438-4639(13)00156-9. doi: 10.1016/j.ijheh.2013.12.001. [Epub ahead of print]
164. He M, Inoue K, Yoshida S, Tanaka M, Takano H, Sun G, Ichinose T. Effects of airway exposure to di-(2-ethylhexyl) phthalate on allergic rhinitis. Immunopharmacol Immunotoxicol. 2013 Jun;35(3):390-5. doi: 10.3109/08923973.2013.787432.
165. Braun JM, Sathyanarayana S, Hauser R. Phthalate exposure and children’s health. Curr Opin Pediatr. 2013 Apr;25(2):247-54. doi: 10.1097/MOP.0b013e32835e1eb6. Review.
166. Hsu NY, Lee CC, Wang JY, Li YC, Chang HW, Chen CY, Bornehag CG, Wu PC, Sundell J, Su HJ. Predicted risk of childhood allergy, asthma, and reported symptoms using measured phthalate exposure in dust and urine. Indoor Air. 2012 Jun;22(3):186-99. doi: 10.1111/j.1600-0668.2011.00753.x. Epub 2011 Nov 16.
167. Park HY, Kim JH, Lim YH, Bae S, Hong YC. Influence of genetic polymorphisms on the association between phthalate exposure and pulmonary function in the elderly. Environ Res. 2013 Apr;122:18-24. doi: 10.1016/j.envres.2012.11.004. Epub 2012 Dec 20.
168. Wu MT, Wu CF, Chen BH, Chen EK, Chen YL, Shiea J, Lee WT, Chao MC, Wu JR. Intake of phthalate-tainted foods alters thyroid functions in Taiwanese children. PLoS One. 2013;8(1):e55005. doi: 10.1371/journal.pone.0055005. Epub 2013 Jan 30.
169. Andra SS, Makris KC. Thyroid disrupting chemicals in plastic additives and thyroid health. J Environ Sci Health C Environ Carcinog Ecotoxicol Rev. 2012;30(2):107-51. doi: 10.1080/10590501.2012.681487. Review.
170. Boas M, Feldt-Rasmussen U, Main KM. Thyroid effects of endocrine disrupting chemicals. Mol Cell Endocrinol. 2012 May 22;355(2):240-8. doi: 10.1016/j.mce.2011.09.005. Epub 2011 Sep 10. Review.
171. Boas M, Feldt-Rasmussen U, Skakkebaek NE, Main KM. Environmental chemicals and thyroid function. Eur J Endocrinol. 2006 May;154(5):599-611. Review.
172. Kobrosly RW, Evans S, Miodovnik A, Barrett ES, Thurston SW, Calafat AM, Swan SH. Prenatal Phthalate Exposures and Neurobehavioral Development Scores in Boys and Girls at 6-10 Years of Age. Environ Health Perspect. 2014 Feb 21. [Epub ahead of print]
173. Szychowski KA, Wójtowicz AK. [Components of plastic disrupt the function of the nervous system]. Postepy Hig Med Dosw (Online). 2013 May 27;67:499-506. Review. Polish.
174. Téllez-Rojo MM, Cantoral A, Cantonwine DE, Schnaas L, Peterson K, Hu H, Meeker JD. Prenatal urinary phthalate metabolites levels and neurodevelopment in children at two and three years of age. Sci Total Environ. 2013 Sep 1;461-462:386-90. doi: 10.1016/j.scitotenv.2013.05.021. Epub 2013 Jun 5.
175. Jurewicz J, Polańska K, Hanke W. Exposure to widespread environmental toxicants and children’s cognitive development and behavioral problems. Int J Occup Med Environ Health. 2013 Apr;26(2):185-204. doi: 10.2478/s13382-013-0099-x. Epub 2013 May 28. Erratum in: Int J Occup Med Environ Health. 2013 Jun;26(3):494.
176. Kim Y, Ha EH, Kim EJ, Park H, Ha M, Kim JH, Hong YC, Chang N, Kim BN. Prenatal exposure to phthalates and infant development at 6 months: prospective Mothers and Children’s Environmental Health (MOCEH) study. Environ Health Perspect. 2011 Oct;119(10):1495-500. doi: 10.1289/ehp.1003178. Epub 2011 Jul 7.
177. Chen FP, Chien MH. Lower concentrations of phthalates induce proliferation in human breast cancer cells. Climacteric. 2013 Dec 27. [Epub ahead of print]
178. Sprague BL, Trentham-Dietz A, Hedman CJ, Wang J, Hemming JD, Hampton JM, Buist DS, Aiello Bowles EJ, Sisney GS, Burnside ES. Circulating serum xenoestrogens and mammographic breast density.
Breast Cancer Res. 2013 May 27;15(3):R45. [Epub ahead of print]
179. Wang YC, Chen HS, Long CY, Tsai CF, Hsieh TH, Hsu CY, Tsai EM. Possible mechanism of phthalates-induced tumorigenesis. Kaohsiung J Med Sci. 2012 Jul;28(7 Suppl):S22-7. doi:10.1016/j.kjms.2012.05.006. Epub 2012 Jul 10. Review.
180. Hsieh TH, Tsai CF, Hsu CY, Kuo PL, Lee JN, Chai CY, Wang SC, Tsai EM. Phthalates induce proliferation and invasiveness of estrogen receptor-negative breast cancer through the AhR/HDAC6/c-Myc signaling pathway. FASEB J. 2012 Feb;26(2):778-87. doi: 10.1096/fj.11-191742. Epub 2011 Nov 2.
181. López-Carrillo L, Hernández-Ramírez RU, Calafat AM, Torres-Sánchez L, Galván-Portillo M, Needham LL, Ruiz-Ramos R, Cebrián ME. Exposure to phthalates and breast cancer risk in northern Mexico. Environ Health Perspect. 2010 Apr;118(4):539-44. doi: 10.1289/ehp.0901091.
182. http://www.nrdc.org/living/chemicalindex/perchlorate.asp
183. http://www.ewg.org/research/rocket-fuel-cows-milk-perchlorate
184. http://www.ewg.org/enviroblog/2008/01/perchlorate-you-might-not-know-how-pronounce-it-its-you
185. http://www.ewg.org/research/rocket-fuel-cows-milk-perchlorate
186. http://www.ewg.org/research/rocket-fuel-cows-milk-perchlorate
188. http://www.ewg.org/research/rocket-fuel-cows-milk-perchlorate
189. Steinmaus C1, Miller MD, Smith AH. Perchlorate in drinking water during pregnancy and neonatal thyroid hormone levels in California. J Occup Environ Med. 2010 Dec;52(12):1217-524. doi: 10.1097/JOM.0b013e3181fd6fa7.
190. Kirk AB. Environmental perchlorate: why it matters. Anal Chim Acta. 2006 May 10;567(1):4-12. Epub 2006 Mar 27.
191. Pearce EN, Braverman LE. Environmental pollutants and the thyroid. Best Pract Res Clin Endocrinol Metab. 2009 Dec;23(6):801-13. doi: 10.1016/j.beem.2009.06.003. Review.
192. Julvez J, Alvarez-Pedrerol M, Rebagliato M, Murcia M, Forns J, Garcia-Esteban R, Lertxundi N, Espada M, Tardón A, Riaño Galán I, Sunyer J. Thyroxine levels during pregnancy in healthy women and early child neurodevelopment. Epidemiology. 2013 Jan;24(1):150-7. doi: 10.1097/EDE.0b013e318276ccd3.
193. Rivkees SA, Mandel SJ. Thyroid disease in pregnancy. Horm Res Paediatr. 2011;76 Suppl 1:91-6. doi: 10.1159/000329186. Epub 2011 Jul 21.
194. LaFranchi SH, Haddow JE, Hollowell JG. Is thyroid inadequacy during gestation a risk factor for adverse pregnancy and developmental outcomes? Thyroid. 2005 Jan;15(1):60-71.
195. Williams GR. Neurodevelopmental and neurophysiological actions of thyroid hormone. J Neuroendocrinol. 2008 Jun;20(6):784-94. doi: 10.1111/j.1365-2826.2008.01733.x. Review.
196. Morreale de Escobar G, Obregon MJ, Escobar del Rey F. Role of thyroid hormone during early brain development. Eur J Endocrinol. 2004 Nov;151 Suppl 3:U25-37. Review.
197. Richard Dahl. Thyroid Alert: Low Iodine and Perchlorate Effects in Women. Environ Health Perspect. 2006 December; 114(12): A714.
198. Brent GA. Braverman LE. Zoeller RT. Thyroid health and the environment. Thyroid. 2007;17:807–809.
199. http://www.ewg.org/research/healthy-home-tips/tip-4-avoid-fire-retardants
200. Kim YR, Harden FA, Toms LM, Norman RE. Health consequences of exposure to brominated flame retardants: A systematic review. Chemosphere. 2014 Feb 11. pii: S0045-6535(13)01729-3. doi: 10.1016/j.chemosphere.2013.12.064. [Epub ahead of print] Review.
201. Springer C, Dere E, Hall SJ, McDonnell EV, Roberts SC, Butt CM, Stapleton HM, Watkins DJ, McClean MD, Webster TF, Schlezinger JJ, Boekelheide K. Rodent thyroid, liver, and fetal testis toxicity of the monoester metabolite of bis-(2-ethylhexyl) tetrabromophthalate (tbph), a novel brominated flame retardant present in indoor dust. Environ Health Perspect. 2012 Dec;120(12):1711-9. doi: 10.1289/ ehp.1204932. Epub 2012 Sep 26.
202. Gundersen Y1, Vaagenes P, Reistad T, Opstad PK. [Brominated flame retardants may cause brain injuries in the fetus and the newborn]. [Article in Norwegian] Tidsskr Nor Laegeforen. 2005 Nov 17;125(22):3098-100.
203. Leijs MM, ten Tusscher GW, Olie K, van Teunenbroek T, van Aalderen WM, de Voogt P, Vulsma T, Bartonova A, Krayer von Krauss M, Mosoiu C, Riojas-Rodriguez H, Calamandrei G, Koppe JG. Thyroid hormone metabolism and environmental chemical exposure. Environ Health. 2012 Jun 28;11 Suppl 1:S10. doi: 10.1186/1476-069X-11-S1-S10.
204. Gill U, Chu I, Ryan JJ, Feeley M. Polybrominated diphenyl ethers: human tissue levels and toxicology. Rev Environ Contam Toxicol. 2004;183:55-97. Review.
205. Wang H, Zhang Y, Liu Q, Wang F, Nie J, Qian Y. Examining the relationship between brominated flame retardants (BFR) exposure and changes of thyroid hormone levels around e-waste dismantling sites. Int J Hyg Environ Health. 2010 Sep;213(5):369-80. doi: 10.1016/j.ijheh.2010.06.004. Epub 2010 Jul 3.
206. Saunders DM, Higley EB, Hecker M, Mankidy R, Giesy JP. In vitro endocrine disruption and TCDD-like effects of three novel brominated flame retardants: TBPH, TBB, & TBCO. Toxicol Lett. 2013 Nov 25;223(2):252-9. doi: 10.1016/j.toxlet.2013.09.009. Epub 2013 Sep 21.
207. Gosavi RA, Knudsen GA, Birnbaum LS, Pedersen LC. Mimicking of estradiol binding by flame retardants and their metabolites: a crystallographic analysis. Environ Health Perspect. 2013 Oct;121(10):1194-9. doi: 10.1289/ehp.1306902. Epub 2013 Aug 13.
208. Johnson PI, Stapleton HM, Mukherjee B, Hauser R, Meeker JD. Associations between brominated flame retardants in house dust and hormone levels in men. Sci Total Environ. 2013 Feb 15;445-446:177-84. doi: 10.1016/j.scitotenv.2012.12.017. Epub 2013 Jan 16.
209. Kiciński M, Viaene MK, Den Hond E, Schoeters G, Covaci A, Dirtu AC, Nelen V, Bruckers L, Croes K, Sioen I, Baeyens W, Van Larebeke N, Nawrot TS. Neurobehavioral function and low-level exposure to brominated flame retardants in adolescents: a cross-sectional study. Environ Health. 2012 Nov 14;11:86. doi: 10.1186/1476-069X-11-86.
210. Roze E, Meijer L, Bakker A, Van Braeckel KN, Sauer PJ, Bos AF. Prenatal exposure to organohalogens, including brominated flame retardants, influences motor, cognitive, and behavioral performance at school age. Environ Health Perspect. 2009 Dec;117(12):1953-8. doi: 10.1289/ehp.0901015. Epub 2009 Aug 31.
211. Dingemans MM, van den Berg M, Westerink RH. Neurotoxicity of brominated flame retardants: (in)direct effects of parent and hydroxylated polybrominated diphenyl ethers on the (developing) nervous system. Environ Health Perspect. 2011 Jul;119(7):900-7. doi: 10.1289/ehp.1003035. Epub 2011 Jan 18. Review.
212. Eriksson P, Fischer C, Fredriksson A. Polybrominated diphenyl ethers, a group of brominated flame retardants, can interact with polychlorinated biphenyls in enhancing developmental neurobehavioral defects. Toxicol Sci. 2006 Dec;94(2):302-9. Epub 2006 Sep 15.
213. Eriksson, P., Jakobsson, E., & Fredriksson, A. Brominated Flame Retardants: A Novel Class of Developmental Neurotoxicants in our Environment?. Environmental Health Perspectives 109(9), 903-908, 2004.
214. Viberg, H., Fredriksson, A., Jakobsson, E., Orn, U., & Eriksson, P. (2003). Neurobehavioral Derangements in Adult Mice Receiving Decabrominated Diphenyl Ether (PBDE 209) During a Defined Period of Neonatal Brain Development. Toxicol Sci 76(1), 112-120.
215. Small CM, Murray D, Terrell ML, Marcus M. Reproductive outcomes among women exposed to a brominated flame retardant in utero. Arch Environ Occup Health. 2011;66(4):201-8. doi: 10.1080/19338244.2010.539640.
216. Jamieson DJ, Terrell ML, Aguocha NN, Small CM, Cameron LL, Marcus M. Dietary exposure to brominated flame retardants and abnormal Pap test results. J Womens Health (Larchmt). 2011 Sep;20(9):1269-78. doi: 10.1089/jwh.2010.2275. Epub 2011 Jul 28.
217. Small CM, DeCaro JJ, Terrell ML, Dominguez C, Cameron LL, Wirth J, Marcus M. Maternal exposure to a brominated flame retardant and genitourinary conditions in male offspring. Environ Health Perspect. 2009 Jul;117(7):1175-9. doi: 10.1289/ehp.0800058. Epub 2009 Feb 27.
218. McGee SP, Konstantinov A, Stapleton HM, Volz DC. Aryl phosphate esters within a major PentaBDE replacement product induce cardiotoxicity in developing zebrafish embryos: potential role of the aryl hydrocarbon receptor. Toxicol Sci. 2013 May;133(1):144-56. doi: 10.1093/toxsci/kft020. Epub 2013 Feb 1.
219. Lim JS, Lee DH, Jacobs DR Jr. Association of brominated flame retardants with diabetes and metabolic syndrome in the U.S. population, 2003-2004. Diabetes Care. 2008 Sep;31(9):1802-7. doi: 10.2337/dc08-0850. Epub 2008 Jun 16.
220. http://www.atsdr.cdc.gov/toxfaqs/tf.asp?id=528&tid=94
221. Akutsu K, Hori S. [Polybrominated diphenyl ether flame retardants in foodstuffs and human milk]. Shokuhin Eiseigaku Zasshi. 2004 Aug;45(4):175-83. Review. Japanese.
222. Schmidt, Charles. PBDEs in breast milk: levels higher in United States than in Europe – Science Selections. Environmental Health Perspectives. Nov 2003.
223. Stiffler, Lisa. “PBDEs: They are everywhere, they accumulate and they spread”. The Seattle Post Intelligentser, March 28, 2007.
224. http://www.ewg.org/research/pbdes-fire-retardants-dust
225. http://www.ewg.org/research/pbdes-fire-retardants-dust
226. http://www.niehs.nih.gov/health/materials/perflourinated_chemicals_508.pdf
227. http://www.ewg.org/research/dirty-dozen-list-endocrine-disruptors
228. http://www.ewg.org/research/dirty-dozen-list-endocrine-disruptors
229. De Coster S, van Larebeke N. Endocrine-disrupting chemicals: associated disorders and mechanisms of action. J Environ Public Health. 2012;2012:713696. Epub 2012 Sep 6. Review.
230. Sally S. White, Suzanne E. Fenton, Erin P. Hines. Endocrine disrupting properties of perfluorooctanoic acid. J Steroid Biochem Mol Biol. Author manuscript; available in PMC 2012 April 24. Published in final edited form as: J Steroid Biochem Mol Biol. 2011 October; 127(1-2): 16–26. Published online 2011 March 21.
231. Kjeldsen LS, Bonefeld-Jørgensen EC. Perfluorinated compounds affect the function of sex hormone receptors. Environ Sci Pollut Res Int. 2013 Nov;20(11):8031-44. doi: 10.1007/s11356-013-1753-3. Epub 2013 Jun 14.
232. Vested A, Ramlau-Hansen CH, Olsen SF, Bonde JP, Kristensen SL, Halldorsson TI, Becher G, Haug LS, Ernst EH, Toft G. Associations of in utero exposure to perfluorinated alkyl acids with human semen quality and reproductive hormones in adult men. Environ Health Perspect. 2013 Apr;121(4):453-8, 458e1-5. doi: 10.1289/ehp.1205118. Epub 2013 Jan 23.
233. Felix Grün, Bruce Blumberg. Endocrine disrupters as obesogens. Mol Cell Endocrinol. Author manuscript; available in PMC 2010 May 25. Published in final edited form as: Mol Cell Endocrinol. 2009 May 25; 304(1-2): 19–29. Published online 2009 March 9. doi: 10.1016/j.mce.2009.02.018
234. Wen LL, Lin LY, Su TC, Chen PC, Lin CY. Association between serum perfluorinated chemicals and thyroid function in U.S. adults: the National Health and Nutrition Examination Survey 2007-2010. J Clin Endocrinol Metab. 2013 Sep;98(9):E1456-64. doi: 10.1210/jc.2013-1282. Epub 2013 Jul 17.
235. Boas M, Feldt-Rasmussen U, Main KM. Thyroid effects of endocrine disrupting chemicals. Mol Cell Endocrinol. 2012 May 22;355(2):240-8. doi: 10.1016/j.mce.2011.09.005. Epub 2011 Sep 10. Review.
236. Marie-Louise Hartoft-Nielsen, Malene Boas, Sofie Bliddal, Åase Krogh Rasmussen, Katharina Main, Ulla Feldt-Rasmussen. Do Thyroid Disrupting Chemicals Influence Foetal Development during Pregnancy? J Thyroid Res. 2011; 2011: 342189. Published online 2011 September 11. doi: 10.4061/2011/342189
237. David Melzer, Neil Rice, Michael H. Depledge, William E. Henley, Tamara S. Galloway. Association between Serum Perfluorooctanoic Acid (PFOA) and Thyroid Disease in the U.S. National Health and Nutrition Examination Survey. Environ Health Perspect. 2010 May; 118(5): 686–692. Published online 2010 January 20. doi: 10.1289/ehp.0901584
238. Corsini E, Luebke RW, Germolec DR, Dewitt JC. Perfluorinated compounds: Emerging POPs with potential immunotoxicity. Toxicol Lett. 2014 Feb 3. pii: S0378-4274(14)00058-7. doi: 10.1016/j.toxlet.2014.01.038. [Epub ahead of print]
239. Wang IJ, Hsieh WS, Chen CY, Fletcher T, Lien GW, Chiang HL, Chiang CF, Wu TN, Chen PC. The effect of prenatal perfluorinated chemicals exposures on pediatric atopy. Environ Res. 2011 Aug;111(6):785-91. doi: 10.1016/j.envres.2011.04.006. Epub 2011 May 23.
240. Liu C, Gin KY, Chang VW. Multi-biomarker responses in green mussels exposed to PFCs: effects at molecular, cellular, and physiological levels. Environ Sci Pollut Res Int. 2014 Feb;21(4):2785-94. doi: 10.1007/s11356-013-2216-6. Epub 2013 Oct 17.
241. Uhl SA, James-Todd T, Bell ML. Association of Osteoarthritis with Perfluorooctanoate and Perfluorooctane Sulfonate in NHANES 2003-2008. Environ Health Perspect. 2013 Apr;121(4):447-52, 452e1-3. doi: 10.1289/ehp.1205673. Epub 2013 Feb 7.
242. Theodore A. Slotkin, Emiko A. MacKillop, Ronald L. Melnick, Kristina A. Thayer, Frederic J. Seidler. Developmental Neurotoxicity of Perfluorinated Chemicals Modeled in Vitro. Environ Health Perspect. 2008 June; 116(6): 716–722. Published online 2008 March 3. doi: 10.1289/ehp.11253
243. Chen Zee E, Cornet P, Lazimi G, Rondet C, Lochard M, Magnier AM, Ibanez G. [Impact of endocrine disrupting chemicals on birth outcomes]. Gynecol Obstet Fertil. 2013 Oct;41(10):601-10. doi: 10.1016/j.gyobfe.2013.08.012. Epub 2013 Oct 9. French.
244. Mei-Huei Chen, Eun-Hee Ha, Ting-Wen Wen, Yi-Ning Su, Guang-Wen Lien, Chia-Yang Chen, Pau-Chung Chen, Wu-Shiun Hsieh. Perfluorinated Compounds in Umbilical Cord Blood and Adverse Birth Outcomes. PLoS One. 2012; 7(8): e42474. Published online 2012 August 3. doi: 10.1371/journal.pone.0042474
245. Fei C, McLaughlin JK, Tarone RE, Olsen J. Perfluorinated chemicals and fetal growth: a study within the Danish National Birth Cohort. Environ Health Perspect. 2007 Nov;115(11):1677-82.
246. De Coster S, van Larebeke N. Endocrine-disrupting chemicals: associated disorders and mechanisms of action. J Environ Public Health. 2012;2012:713696. Epub 2012 Sep 6. Review.
247. Mireia Gascon, Marta Fort, David Martínez, Anne-Elie Carsin, Joan Forns, Joan O. Grimalt, Loreto Santa Marina, Nerea Lertxundi, Jordi Sunyer, Martine Vrijheid. Polybrominated Diphenyl Ethers (PBDEs) in Breast Milk and Neuropsychological Development in Infants. Environ Health Perspect. 2012 December; 120(12): 1760–1765. Published online 2012 September 25. doi: 10.1289/ehp.1205266
248. Wargo, John. Our Children’s Toxic Legacy: How Science and Law Fail to Protect Us from Pesticides. Yale University Press. 2nd edition (1998), page 3.
249. Wargo, John. Our Children’s Toxic Legacy: How Science and Law Fail to Protect Us from Pesticides. Yale University Press. 2nd edition (1998), pages 11-12.
250. Carol J. Burns, Laura J. McIntosh, Pamela J. Mink, Anne M. Jurek, Abby A. Li. Pesticide Exposure and Neurodevelopmental Outcomes: Review of the Epidemiologic and Animal Studies. J Toxicol Environ Health B Crit Rev. 2013 April; 16(3-4): 127–283. Published online 2013 June 18. doi: 10.1080/10937404.2013.783383
251. National Academy of Sciences. Research on children and adolescents with mental, behavioral, and developmental disorders. Washington, DC: National Academy Press; 1988.
252. Wargo, John. Our Children’s Toxic Legacy: How Science and Law Fail to Protect Us from Pesticides. Yale University Press. 2nd edition (1998) 402 pages.
253. Leslie London, Cheryl Beseler, Maryse F. Bouchard, David C. Bellinger, Claudio Colosio, Philippe Grandjean, Raul Harari, Tahira Kootbodien, Hans Kromhout, Francesca Little, Tim Meijster, Angelo Moretto, Diane S. Rohlman, Lorann Stallones. Neurobehavioural and neurodevelopmental effects of pesticide exposures. Neurotoxicology. Author manuscript; available in PMC 2013 August 1. Published in final edited form as: Neurotoxicology. 2012 August; 33(4): 887–896. Published online 2012 January 17. doi: 10.1016/j.neuro.2012.01.004
254. Raul Harari, Jordi Julvez, Katsuyuki Murata, Dana Barr, David C. Bellinger, Frodi Debes, Philippe Grandjean. Neurobehavioral Deficits and Increased Blood Pressure in School-Age Children Prenatally Exposed to Pesticides. Environ Health Perspect. 2010 June; 118(6): 890–896. Published online 2010 February 25. doi: 10.1289/ehp.0901582
257. Devon Payne-Sturges, Jonathan Cohen, Rosemary Castorina, Daniel A. Axelrad, Tracey J. Woodruff. Evaluating Cumulative OP Pesticide Body Burden of Children: A National Case Study. Environ Sci Technol. Author manuscript; available in PMC 2010 October 15. Published in final edited form as: Environ Sci Technol. 2009 October 15; 43(20): 7924–7930. doi: 10.1021/es900713s
258. Maryse F. Bouchard, David C. Bellinger, Robert O. Wright, Marc G. Weisskopf. Attention deficit/hyperactivity disorder and urinary metabolites of organophosphate pesticides in U.S. children 8–15 years. Pediatrics. Author manuscript; available in PMC 2013 July 9. Published in final edited form as: Pediatrics. 2010 June; 125(6): e1270–e1277. Published online 2010 May 17. doi: 10.1542/peds.2009-3058
259. Curl C. L. Fenske R. A. Kissel J. C. Shirai J. H. Moate T. F. Griffith W. Coronado G. Thompson B. Evaluation of take-home organophosphorus pesticide exposure among agricultural workers and their children. Environ. Health Perspect. 2002;110:A787–A792.
260. Fenske R. A. Lu C. Barr D. Needham L. Children’s exposure to chlorpyrifos and parathion in an agricultural community in central Washington State. Environ. Health Perspect. 2002;110:549–553.
261. Curwin B. D. Hein M. J. Sanderson W. T. Striley C. Heederik D. Kromhout H. Reynolds S. J. Alavanja M. C. Urinary pesticide concentrations among children, mothers and fathers living in farm and non-farm households in Iowa. Ann. Occup. Hyg. 2007;51:53–65.
262. Arcury T. A. Quandt S. A. Barr D. B. Hoppin J. A. Mccauley L. Grzywacz J. G. Robson M. G. Farmworker exposure to pesticides: Methodologic issues for the collection of comparable data. Environ. Health Perspect. 2006;114:923–928.
266. Starek A, Szabla J. [Ethylene glycol alkyl ethers–the substances noxious to health]. Med Pr. 2008;59(2): 179-85. Review. Polish.
267. Cordier S, Garlantézec R, Bonvallot N, Multigner L. Glycol ethers and congenital malformations.
Epidemiology. 2013 Nov;24(6):940. doi: 10.1097/EDE.0b013e3182a79ac7. No abstract available.
268. El-Zein RA, Abdel-Rahman SZ, Morris DL, Legator MS. Exposure to ethylene glycol monomethyl ether: clinical and cytogenetic findings. Arch Environ Health. 2002 Jul-Aug;57(4):371-6.
269. Cherry N, Moore H, McNamee R, Pacey A, Burgess G, Clyma JA, Dippnall M, Baillie H, Povey A; participating centres of Chaps-UK. Occupation and male infertility: glycol ethers and other exposures. Occup Environ Med. 2008 Oct;65(10):708-14. doi: 10.1136/oem.2007.035824. Epub 2008 Apr 16.
270. Cicolella A. [Glycol ethers reproductive risks]. Gynecol Obstet Fertil. 2006 Oct;34(10):955-63. Epub 2006 Sep 20. Review. French.
271. Jensen TK, Bonde JP, Joffe M. The influence of occupational exposure on male reproductive function. Occup Med (Lond). 2006 Dec;56(8):544-53. Review.
272. Sheiner EK, Sheiner E, Hammel RD, Potashnik G, Carel R. Effect of occupational exposures on male fertility: literature review. Ind Health. 2003 Apr;41(2):55-62. Review.
273. Massaad C, Entezami F, Massade L, Benahmed M, Olivennes F, Barouki R, Hamamah S. How can chemical compounds alter human fertility? Eur J Obstet Gynecol Reprod Biol. 2002 Jan 10;100(2):127-37. Review.
274. Adedara IA, Farombi EO: Induction of oxidative damage in the testes and spermatozoa and hematotoxicity in rats exposed to multiple doses of ethylene glycol monoethyl ether. Hum Exp Toxicol. 2010, 29, 801–812.
275. Bagchi G, Waxman DJ. Toxicity of ethylene glycol monomethyl ether: impact on testicular gene expression. Int J Androl. 2008, 31, 269–274.
276. Multigner L, Ben Brik E, Arnaud I, Haguenoer JM, Jouannet P, Auger J, Eustache F. Glycol ethers and semen quality: a cross-sectional study among male workers in the Paris Municipality. Occup Environ Med. 2007 Jul;64(7):467-73. Epub 2007 Mar 1.
277. Welch LS, Schrader SM, Turner TW, Cullen MR. Effects of exposure to ethylene glycol ethers on shipyard painters II. Male reproduction. Am J Ind Med. 1988, 14, 509–526.
278. Hsieh GY, Wang JD, Cheng TJ, Chen PC. Prolonged menstrual cycles in female workers exposed to ethylene glycol ethers in the semiconductor manufacturing industry. Occup Environ Med. 2005 Aug;62(8):510-6.
279. Chen PC, Hsieh GY, Wang JD, Cheng TJ. Prolonged time to pregnancy in female workers exposed to ethylene glycol ethers in semiconductor manufacturing. Epidemiology. 2002 Mar;13(2):191-6.
Hematological effects
280. Gold EB, Eskenazi B, Hammond SK, Lasley BL, Samuels SJ, O’Neill Rasor M, Hines CJ et al.: Prospectively assessed menstrual cycle characteristics in female wafer-fabrication and nonfabrication semiconductor employees. Am J Ind Med. 1995, 28, 799–815.
281. Davis BJ, Almekinder JL, Flagler N, Travlos G, Wilson R, Maronpot RR. Ovarian luteal cell toxicity of ethylene glycol monomethyl ether and methoxy acetic acid in vivo and in vitro. Toxicol Appl Pharmacol. 1997, 142, 328–337.
282. Shih TS, Hsieh AT, Liao GD, Chen YH, Liou SH. Haematological and spermatotoxic effects of ethylene glycol monomethyl ether in copper clad laminate factories. Occup Environ Med. 2000 May;57(5):348-52.
283. Starek A, Szymczak W, Zapor L. Hematological effects of four ethylene glycol monoalkyl ethers in short-term repeated exposure in rats. Arch Toxicol. 2008, 82, 125–136.
284. Starek-OEwiechowicz B, Miranowicz-Dzier¿awska K, SzymczakW, Budziszewska B, Starek A. Hematological effects of exposure to mixtures of selected ethylene glycol alkyl ethers in rats. Pharmacol Rep. 2012, 64, 166–178.
285. R R Miller, E A Hermann, J T Young, T D Landry, L L Calhoun. Ethylene glycol monomethyl ether and propylene glycol monomethyl ether: metabolism, disposition, and subchronic inhalation toxicity studies. Environ Health Perspect. 1984 August; 57: 233–239.
286. MortonW. Occupational phenoxyethanol neurotoxicity: a report of three cases. J Occup Med. 1990, 32, 42–45.
287. Pomierny B, Starek A, Krzyżanowska W, Starek-Swiechowicz B, Smaga I, Pomierny-Chamioło L, Regulska M, Budziszewska B. Potential neurotoxic effect of ethylene glycol ethers mixtures. Pharmacol Rep. 2013;65(5):1415-21.
288. http://www.ewg.org/news/news-releases/2012/02/10/epa-lists-dry-cleaning-chemical-likely-carcinogen
289. http://www.ewg.org/enviroblog/2011/09/dry-cleaning-chemicals-hang-around-your-clothes
290. Ruder AM. Potential health effects of occupational chlorinated solvent exposure. Ann N Y Acad Sci. 2006 Sep;1076:207-27.
291. Diodovich C, Ferrario D, Casati B, Malerba I, Marafante E, Parent-Massin D, Gribaldo L. Sensitivity of human cord blood cells to tetrachloroethylene: cellular and molecular endpoints. Arch Toxicol. 2005 Sep;79(9):508-14. Epub 2005 Jul 12.
292. Vlaanderen J, Straif K, Ruder A, Blair A, Hansen J, Lynge E, Charbotel B, Loomis D, Kauppinen T, Kyyronen P, Pukkala E, Weiderpass E, Guha N. Tetrachloroethylene Exposure and Bladder Cancer Risk: A Meta-Analysis of Dry-Cleaning-Worker Studies. Environ Health Perspect. 2014 Mar 21. [Epub ahead of print]
293. Guyton KZ, Hogan KA, Scott CS, Cooper GS, Bale AS, Kopylev L, Barone S, Makris SL, Glenn B, Subramaniam RP, Gwinn MR, Dzubow RC, Chiu WA. Human health effects of tetrachloroethylene: key findings and scientific issues. Environ Health Perspect. 2014 Apr;122(4):325-34. doi: 10.1289/ehp.1307359. Epub 2014 Feb 11.
294. Calvert GM, Ruder AM, Petersen MR. Mortality and end-stage renal disease incidence among dry cleaning workers. Occup Environ Med. 2011 Oct;68(10):709-16. doi: 10.1136/oem.2010.060665. Epub 2010 Dec 16.
295. Ma J, Lessner L, Schreiber J, Carpenter DO. Association between residential proximity to PERC dry cleaning establishments and kidney cancer in New York City. J Environ Public Health. 2009;2009:183920. doi: 10.1155/2009/183920. Epub 2010 Jan 24.
296. Guyton KZ, Hogan KA, Scott CS, Cooper GS, Bale AS, Kopylev L, Barone S, Makris SL, Glenn B, Subramaniam RP, Gwinn MR, Dzubow RC, Chiu WA. Human health effects of tetrachloroethylene: key findings and scientific issues. Environ Health Perspect. 2014 Apr;122(4):325-34. doi: 10.1289/ehp.1307359. Epub 2014 Feb 11.
297. Grandjean P, Landrigan PJ. Lancet Neurol. 2014 Mar;13(3):330-8. doi: 10.1016/S1474-4422(13)70278-3. Epub 2014 Feb 17. Review.
298. Aschengrau A, Weinberg JM, Janulewicz PA, Romano ME, Gallagher LG, Winter MR, Martin BR, Vieira VM, Webster TF, White RF, Ozonoff DM. Environ Health. 2012 Jan 20;11:2. doi: 10.1186/1476-069X-11-2.
299. Aschengrau A, Weinberg JM, Janulewicz PA, Gallagher LG, Winter MR, Vieira VM, Webster TF, Ozonoff DM. Prenatal exposure to tetrachloroethylene-contaminated drinking water and the risk of congenital anomalies: a retrospective cohort study. Environ Health. 2009 Sep 24;8:44. doi: 10.1186/1476-069X-8-44.
300. http://www.ewg.org/enviroblog/2011/09/dry-cleaning-chemicals-hang-around-your-clothes
302a. “Formaldehyde in Clothing and Other Textiles” (PDF). Existing Chemicals Information Sheet. Australian National Industrial Chemicals Notification and Assessment Scheme. October 2007.
302b. http://www.ewg.org/enviroblog/2014/01/chemicals-should-disappear-cosmetics
303. Economic Importance, Formaldehyde Council. 2009.
304. http://www.epa.gov/ttn/atw/nata2005/05pdf/sum_results.pdf
305. National Toxicology Program (10 June 2011). “12th Report on Carcinogens”. National Toxicology Program. Retrieved 2011-06-11.
306. Kim KH, Jahan SA, Lee JT. Exposure to formaldehyde and its potential human health hazards. J Environ Sci Health C Environ Carcinog Ecotoxicol Rev. 2011 Oct;29(4):277-99. doi: 10.1080/10590501.2011.629972. Review.
307. Formaldehyde, A CICAD (Concise International Chemical Assessment Document) No. 40. International Programme on Chemical Safety (IPSC). World Health Organization. 2003.
308. “Agency for Toxic Substance & Disease Registry/Environmental Protection Agency”. In Toxicological Profile for Formaldehyde, 468–476. Dept. of Health and Human Services. July 1999.
309. Kulle, T J. 1993. Acute odor and irritation response in healthy nonsmokers with formaldehyde exposure. Inhal Toxicol., 5: 323–332.
310. Zhang, D Z, Zhang, F L, Jin, S Y and Liu, Y H. 1999. Investigation on the health of workers occupationally exposed to low level of formaldehyde. Chinese J Ind Hyg Occup Dis., 17(5): 13–14.
311. Kulle, T J. 1993. Acute odor and irritation response in healthy nonsmokers with formaldehyde exposure. Inhal Toxicol., 5: 323–332.
312. Salonen, H, Pasanen, A L, Lappalainen, S, Riuttala, H, Tuomi, T and Pasanen, P. 2009. Volatile organic compounds and formaldehyde as explaining factors for sensory irritation in office environments. J Occup Environ Hyg, 6(4): 239–247.
313. Cronin, E. 1991. Formaldehyde is a significant allergen in women with hand eczema. Contact Dermatitis, 25(5): 276–282.
314. Meding, B and Swanbeck, G. 1990. Occupational hazard in an industrial city. Contact Dermatitis, 22(1): 13–23.
315. Scheman, A J, Carroll, P A, Brown, K H and Osburn, A H. 1998. Formaldehyde-related textile allergy: An update. Contact Dermatitis, 38(6): 332–336.
316. Rastogi, S C. 1992. A survey of formaldehyde in shampoos and skin creams on the Danish market. Contact Dermatitis, 27(4): 235–240.
317. Su, H and Zheng, R. 1995. Four contact dermatitis cases exposed to phenolic aldehyde resin. J Preven Med, 1: 1–8.
318. Wu, D F and Wu, Y H. 2001. Investigation of dermatitis caused by occupational formaldehyde exposure. J Labour Med, 18(4): 226–227.
319. Brooks, S M, Hammad, Y, Richards, I, Giovonco-Barbas, J and Jenkins, K. 1998. The spectrum of irritant-induced asthma: sudden and not-so sudden onset and the role of allergy. Chest, 113: 42–49.
320. Vandenplas, O, Fievez, P, Delwiche, J P, Boulanger, J and Thimpont, J. 2004. Persistent asthma following accidental exposure to formaldehyde. Allergy, 59: 115–116.
321. Yue, W, Jin, X B, Pan, X C and Ding, J. 2004. Relationship between indoor air formaldehyde exposure and allergic asthma in adults. Chinese J Pub Heal. 20(8): 904–906.
322. Kim, C W, Song, J S, Ahn, Y S, Park, S H, Noh, J H and Hong, C S. 2001. Occupational asthma due to formaldehyde. Yonsei Med J, 42: 440–445.
323. Rumchev, K B, Spickett, J T, Bulsara, M K, Phillips, M R and Stick, S M. 2002. Domestic exposure to formaldehyde significantly increases the risk of asthma in young children. Eur Resp J., 20(2): 403–408.
324. Casset, A, Marchand, C, Purohit, A, le-Calve, S, Uring-Lambert, B and Donnay, C. 2006. Inhaled formaldehyde exposure: Effect on bronchial response to mite allergen in sensitized asthma patients. Allergy, 61: 1344–1350.
325. McGwin, G, Lienert, J and Kennedy, J I. 2010. Formaldehyde exposure and asthma in children: A systematic review. Environ Health Perspect, 118(3): 313–317.
326. Vandenplas, O, Fievez, P, Delwiche, J P, Boulanger, J and Thimpont, J. 2004. Persistent asthma following accidental exposure to formaldehyde. Allergy, 59: 115–116.
327. Krakowiak, A, Gorski, P, Pazdrak, K and Ruta, U. 1998. Airway response to formaldehyde inhalation in asthmatic subjects with suspected respiratory formaldehyde sensitization. Am J Indus Med., 33: 274–281.
328. Dai, D and Bao, Z. 1999. Investigative report on formaldehyde occupational workers. Ind Heal Occup Dis., 1: 43–51.
329. Costa, S, Pina, C, Coelho, P, Costa, C, Silva, SPorto, B. 2011. Occupational exposure to formaldehyde: Genotoxic risk evaluation by comet assay and micronucleus test using human peripheral lymphocytes. J Toxicol Environ Health A, 74: 1040–1051.
330. Li, Z G and Chen, B C. 2002. Effect of low concentration formaldehyde on the health of workers. Chinese J Ind Med., 15(5): 302–303.
331. Geng, Y, Meng, X, Li, X and Lu, G. 2004. Occupational damage in densified wood board producing field and its effect on workers’ health. Occup Heal., 20(8): 21–22.
332. Fan, W, Zhou, Y, Jin, F, Du, L and Jin, X. 2006. The health effects of pathologists exposed to formaldehyde. J Occup Environ Med., 23(6): 466–468.
333. Rager, J E, Smeester, L, Jaspers, I, Sexton, K G and Fry, R C. 2011. Epigenetic changes induced by air toxics: Formaldehyde exposure alters miRNA expression profiles in human lung cells. Environ Health Perspect, 119(4): 494–500.
334. International Agency for Research on Cancer (IARC). 1995. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Wood Dust and Formaldehyde. Lyon, France: International Association for Research on Cancer.
335. Feng, Y, Wang, W, Jiang, Z, Hu, G, Zhong, S and Zhang, H. 1996. Health status of wood workers exposed to formaldehyde. Anhui J Preven Med., 2(2): 99–100.
336. Tang, L X and Zhang, Y S. 2003. Health investigation on workers exposed to formaldehyde. Occup Heal., 19(7): 34–35.
337. Lu, Y, Chen, X J, Yang, X Y and Xue, Z Q. 2007. A survey of the effection to teachers’ health from formaldehyde contact. J Xinjiang Med Uni., 30(3): 234–237.
338. Songur, A, Ozen, O A and Sarsilmaz, M. 2010. The toxic effects of formaldehyde on the nervous system. Rev Environ Contam Toxicol, 203: 105–118.
339. International Agency for Research on Cancer (IARC). 1995. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Wood Dust and Formaldehyde. Lyon, France: International Association for Research on Cancer.
340. Armon, C. 2009. Smoking may be considered an established risk factor for sporadic ALS. Neurology, 73: 1693–1698.
341. Weisskopf, M G, Morozova, N, O’Reilly, E J, McCullough, M L, Calle, E E, Thun, M J and Ascherio, A. 2009. Prospective study of chemical exposures and amyotrophic lateral sclerosis. J Neurol Neurosurg Psychiatry., 80: 558–561.
342. Hong, Z, Tong, Z and Shi, J. 2007. Effects of formaldehyde on respiratory system and pulmonary function of workers. Chinese J Pub Heal., 23(7): 849–850.
343. Kriebel, D, Sama, S R and Cocanour, B. 1993. Reversible pulmonary responses to formaldehyde. A study of clinical anatomy students. Am Rev Respirat Dis, 148: 1509–1515.
344. Akbar-Khanzadeh, F and Mlynek, J S. 1997. Changes in respiratory function after one and three hours of exposure to formaldehyde in non-smoking subjects. Occup Environ Med, 54: 296–300.
345. Wang, W, Wang, Q and Zhou, Y. 2000. Effects of low concentration of formaldehyde on respiratory system and pulmonary function of workers. Chinese J Ind Med., 13(2): 115–116.
346. Li, Z G and Chen, B C. 2002. Effect of low concentration formaldehyde on the health of workers. Chinese J Ind Med., 15(5): 302–303.
347. Fan, W, Zhou, Y, Jin, F, Du, L and Jin, X. 2006. The health effects of pathologists exposed to formaldehyde. J Occup Environ Med., 23(6): 466–468.
348. Tang, L X and Zhang, Y S. 2003. Health investigation on workers exposed to formaldehyde. Occup Heal., 19(7): 34–35.
349. Tong, Z M, Zhu, S X and Shi, J. 2007. Effect of formaldehyde on blood component and blood biochemistry of exposed workers. Chinese J Ind Med., 20(6): 409–410.
350. Yang, W H. 2007. Hemogram of workers exposed to low concentration of formaldehyde. Prac Preven Med., 14(3): 792–799.
351. Huang, Y, Zou, Z and Deng, H. 2007. An analysis report about peripheral blood anemia induced by excessive formaldehyde in abiding place. Jiangsu Environ Sci Technol, 20: 16–17.
352. Huang, W, Lu, Y, Chang, X, Bai, L, Zhao, H and Liu, W. 2007. Indoor air pollution in newly redecorated rooms. J Environ Heal, 24(2): 101–103.
353. Kuo, H, Jian, G, Chen, C, Liu, C and Lai, J. 1997. White blood cell count as an indicator of formaldehyde exposure. Bull Environ Cont Toxicol, 59(2): 261–267.
354. Xu, S Y, Yi, G L and Li, S H. 2007. Hygienic investigation of the effect of formaldehyde on the workers’ health. Occup Heal., 23(7): 491–492.
355. Lu, Y, Chen, X J, Yang, X Y and Xue, Z Q. 2007. A survey of the effection to teachers’ health from formaldehyde contact. J Xinjiang Med Uni., 30(3): 234–237.
356. Taskinen, H K, Kyyronen, P, Hemminki, K, Hoikkala, M, Lajunen, K and Lindbohm, M L. 1994. Laboratory work and pregnancy outcome. J Occup Med., 36(3): 311–319.
357. Taskinen, H K, Kyyronen, P, Sallmen, M, Virtanen, S V, Liukkonen, T A and Huida, O. 1999. Reduced fertility among female wood workers exposed to formaldehyde. Am J Ind Med., 36(1): 206–212.
358. Zhang, L, Steinmaus, C, Eastmond, D A, Xin, X K and Smith, M T. 2009. Formaldehyde exposure and leukemia: A new meta-analysis and potential mechanisms. Muta Res, 681: 150–168.
359. Washam, C. 2011. Epigenetics of formaldehyde: Altered microRNAs may be key to adverse effects. Environ Health Perspect, 119: 176–182.
360. IARC. 2006. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. Vol. 88 Formaldehyde, 2-Butoxyethanol and 1-tert-Butoxypropan-2-ol. Available at: http://monographs.iarc.fr/ENG/Monographs/vol88/index.php
361. Yang, Y, Allen, B C, Tan, Y M, Liao, K H and Clewell, H J. 2010. Bayesian analysis of a rat formaldehyde DNA-protein cross-link model. J Toxicol Environ Health A, 73(12): 787–806.
362. Jiang, L. 2006. Mensuration of indoor formaldehyde pollution of Pingdingshan municipal office building. J Pingdingshan Ins Technol., 15(3): 38–39.
363. Tong, Z M, Shi, J, Zhao, J S, Yang, H, Jiang, R M and Kong, L. 2006. Analysis on genetic toxicity of formaldehyde on occupational exposure population. Chinese J Pub Heal., 22(7): 783–784.
364. Yu, L Q, Jiang, S F, Leng, S G, Zhang, C Z, Yan, Y J and Niu, Y. 2005. Early genetic effects on workers occupationally exposed to formaldehyde. Chinese J Preven Med., 39(6): 392–395.
365. Costa, S, Coelho, P, Costa, C, Silva, S, Mayan, O and Santos, L S. 2008. Genotoxic damage in pathology anatomy laboratory workers exposed to formaldehyde. Toxicology, 252: 40–48.
366. Environment Canada/Health Canada. Canadian Environmental Protection Act. 1999. Priority substances list assessment report. Formaldehyde. Government of Canada, Environment Canada, Health Canada. 2001.
367. World Health Organization. 2002. Concise International Chemical Assessment Document 40. Formaldehyde, Geneva: Author.
368. IARC. 2006. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans, Vol. 88 Formaldehyde, 2-Butoxyethanol and 1-tert-Butoxypropan-2-ol Available at: http://monographs.iarc.fr/ENG/Monographs/vol88/index.php
369. Kim KH, Jahan SA, Lee JT. Exposure to formaldehyde and its potential human health hazards. J Environ Sci Health C Environ Carcinog Ecotoxicol Rev. 2011 Oct;29(4):277-99. doi: 10.1080/10590501.2011.629972. Review.
370. 2011. “National Toxicology Program (NTP)”. In Report on Carcinogens , 12th ed, Department of Health and Human Services, Public Health Service, National Toxicology Program. Available at: http://ntp.niehs.nih.gov/go/roc12
371. Integrated Risk Information System. 2010. Formaldehyde, Washington, DC: US Environmental Protection Agency. Available at: http://cfpub.epa.gov/ncea/iris_drafts/recordisplay.cfm?deid=223614
372. Thompson, C M and Grafstrom, R C. 2011. Considerations for the implausibility of leukemia induction by formaldehyde. Toxicol Sci, 120: 230–232.
373. Schwilk, E, Zhang, L, Smith, M T, Smith, A H and Steinmaus, C. 2010. Formaldehyde and leukemia: An updated meta-analysis and evaluation of bias. J Occup Environ Med, 52: 878–886.
374. Pinkerton, L, Hein, M and Stayner, L. 2004. Mortality among a cohort of garment workers exposed to formaldehyde: An update. Occup Environ Med, 61(3): 193–200.
375. Dreyfuss, J H. 2010. Occupational formaldehyde exposure linked to increased risk of myeloid leukemia and death. CA Cancer J Clin, 60(3): 135–136.
376. Crinnion WJ. Toxic effects of the easily avoidable phthalates and parabens. Altern Med Rev. 2010 Sep;15(3):190-6. Review.
377. Ye X, Bishop AM, Reidy JA, Needham LL, Calafat AM. Parabens as urinary biomarkers of exposure in humans. Environ Health Perspect. 2006 Dec;114(12):1843-6.
379. Canosa P, Rodríguez I, Rubí E, Cela R. Determination of parabens and triclosan in indoor dust using matrix solid-phase dispersion and gas chromatography with tandem mass spectrometry. Anal Chem. 2007 Feb 15;79(4):1675-81.
380. Crinnion WJ. Toxic effects of the easily avoidable phthalates and parabens. Altern Med Rev. 2010 Sep;15(3):190-6. Review.
381. Darbre PD, Harvey PW. Paraben esters: review of recent studies of endocrine toxicity, absorption, esterase and human exposure, and discussion of potential human health risks. J Appl Toxicol. 2008 Jul;28(5):561-78. doi: 10.1002/jat.1358. Review.
382. Cheng S, Leow YH, Goh CL, Goon A. Contact sensitivity to preservatives in Singapore: frequency of sensitization to 11 common preservatives 2006-2011. Dermatitis. 2014 Mar-Apr;25(2):77-82. doi: 10.1097/DER.0000000000000031.
383. Travassos AR, Claes L, Boey L, Drieghe J, Goossens A. Non-fragrance allergens in specific cosmetic products. Contact Dermatitis. 2011 Nov;65(5):276-85. doi: 10.1111/j.1600-0536.2011.01968.x. Epub 2011, Sep 21.
384. Soni MG, Taylor SL, Greenberg NA, Burdock GA. Evaluation of the health aspects of methyl paraben: a review of the published literature. Food Chem Toxicol. 2002 Oct;40(10):1335-73. Review.
385. Soni MG, Burdock GA, Taylor SL, Greenberg NA. Safety assessment of propyl paraben: a review of the published literature. Food Chem Toxicol. 2001 Jun;39(6):513-32. Review.
386. Mowad CM. Allergic contact dermatitis caused by parabens: 2 case reports and a review. Am J Contact Dermat. 2000 Mar;11(1):53-6. Review.
387. Darbre PD, Harvey PW. Paraben esters: review of recent studies of endocrine toxicity, absorption, esterase and human exposure, and discussion of potential human health risks. J Appl Toxicol. 2008 Jul;28(5):561-78. doi: 10.1002/jat.1358. Review.
388. Ge JH, Chang B. [Estrogenic activities of parabens]. Wei Sheng Yan Jiu. 2006 Sep;35(5):650-2. Review. Chinese.
389. Scialli AR. Reproductive effects of the parabens. Reprod Toxicol. 2011 Jul;32(1):138-40; author reply 141. doi: 10.1016/j.reprotox.2011.03.005. Epub 2011 Mar 31.
390. Boberg J, Taxvig C, Christiansen S, Hass U. Possible endocrine disrupting effects of parabens and their metabolites. Reprod Toxicol. 2010 Sep; 30(2):301-12. Epub 2010 Apr 8.
391. Kim TS, Kim CY, Lee HK, Kang IH, Kim MG, Jung KK, Kwon YK, Nam HS, Hong SK, Kim HS, Yoon HJ, Rhee GS. Estrogenic Activity of Persistent Organic Pollutants and Parabens Based on the Stably Transfected Human Estrogen Receptor-α Transcriptional Activation Assay (OECD TG 455). Toxicol Res. 2011 Sep;27(3):181-4. doi: 10.5487/TR.2011.27.3.181.
392. Vos J.G., Dybing E., Greim H.A., Ladefoged O., Lambre C., Tarazona J.V., Brandt I., Vethaak A.D. Health effects of endocrine-disrupting chemicals on wildlife, with special reference to the European situation. Crit. Rev. Toxicol. (2000);30:71–133.
393. Christiansen S, Kortenkamp A, Axelstad M, Boberg J, Scholze M, Jacobsen PR, Faust M, Lichtensteiger W, Schlumpf M, Burdorf A, Hass U. Mixtures of endocrine disrupting contaminants modelled on human high end exposures: an exploratory study in rats. Int J Androl. 2012 Jun;35(3):303-16. doi: 10.1111/j.1365-2605.2011.01242.x. Epub 2012 Feb 28.
394. Kim SM, Jung EM, An BS, Hwang I, Vo TT, Kim SR, Lee SM, Choi KC, Jeung EB. Additional effects of bisphenol A and paraben on the induction of calbindin-D(9K) and progesterone receptor via an estrogen receptor pathway in rat pituitary GH3 cells. J Physiol Pharmacol. 2012 Oct;63(5):445-55.
395. Ahn HJ, An BS, Jung EM, Yang H, Choi KC, Jeung EB. Parabens inhibit the early phase of folliculogenesis and steroidogenesis in the ovaries of neonatal rats. Mol Reprod Dev. 2012 Sep;79(9):626-36. doi: 10.1002/mrd.22070. Epub 2012 Jul 26.
396. Smith KW, Souter I, Dimitriadis I, Ehrlich S, Williams PL, Calafat AM, Hauser R. Urinary paraben concentrations and ovarian aging among women from a fertility center. Environ Health Perspect. 2013 Nov-Dec;121(11-12):1299-305. doi: 10.1289/ehp.1205350. Epub 2013 Aug 1.
397. Towers CV, Chambers W, Lewis D, Howard B, Chen J, Terry P. Transplacental passage of antimicrobial paraben preservatives. Obstet Gynecol. 2014 May;123 Suppl 1:175S-6S. doi: 10.1097/01. AOG.0000447188.44313.6e.
398. Konduracka E, Krzemieniecki K, Gajos G. Relationship between everyday use cosmetics and female breast cancer. Pol Arch Med Wewn. 2014 Apr 2. pii: AOP_14_026. [Epub ahead of print]
399. Błędzka D, Gromadzińska J, Wąsowicz W. Parabens. From environmental studies to human health. Environ Int. 2014 Jun;67:27-42. doi: 10.1016/j.envint.2014.02.007. Epub 2014 Mar 19. Review.
400. Darbre PD, Harvey PW. Paraben esters: review of recent studies of endocrine toxicity, absorption, esterase and human exposure, and discussion of potential human health risks. J Appl Toxicol. 2008 Jul;28(5):561-78. doi: 10.1002/jat.1358. Review.
401. Darbre PD. Environmental oestrogens, cosmetics and breast cancer. Best Pract Res Clin Endocrinol Metab. 2006 Mar;20(1):121-43. Review.
402. Harvey PW, Darbre P. Endocrine disrupters and human health: could oestrogenic chemicals in body care cosmetics adversely affect breast cancer incidence in women? J Appl Toxicol. 2004 May-Jun;24(3):167-76. Review.
403. Darbre PD, Aljarrah A, Miller WR, Coldham NG, Sauer MJ, Pope GS. Concentrations of parabens in human breast tumours. J Appl Toxicol. 2004 Jan-Feb;24(1):5-13.
404. Charles AK, Darbre PD. Combinations of parabens at concentrations measured in human breast tissue can increase proliferation of MCF-7 human breast cancer cells. J Appl Toxicol. 2013 May;33(5):390-8. doi: 10.1002/jat.2850. Epub 2013 Jan 31.
405. Vo TT1, Jeung EB. An evaluation of estrogenic activity of parabens using uterine calbindin-d9k gene in an immature rat model. Toxicol Sci. 2009 Nov;112(1):68-77. doi:10.1093/toxsci/kfp176. Epub 2009 Aug 4.
406. Pugazhendhi D, Pope GS, Darbre PD. Oestrogenic activity of p-hydroxybenzoic acid (common metabolite of paraben esters) and methylparaben in human breast cancer cell lines. J Appl Toxicol. 2005 Jul-Aug;25(4):301-9.
407. Okubo T., Yokoyama Y., Kano K., Kano I. ERdependent estrogenic activity of parabens assessed by proliferation of human breast cancer MCF-7 cells and expression of ERalpha and PR. Food Chem. Toxicol. (2001);39:1225–1232. doi: 10.1016/S0278-6915(01)00073-4.
408. Khanna S, Dash PR, Darbre PD. Exposure to parabens at the concentration of maximal proliferative response increases migratory and invasive activity of human breast cancer cells in vitro. J Appl Toxicol. 2014 Mar 20. doi: 10.1002/jat.3003. [Epub ahead of print]
410. http://www.ewg.org/enviroblog/2014/01/chemicals-should-disappear-cosmetics
412. http://www.fda.gov/forconsumers/consumerupdates/ucm205999.htm
413. Tan L, Nielsen NH, Young DC, Trizna Z; Council on Scientific Affairs, American Medical Association. Use of antimicrobial agents in consumer products. Arch Dermatol. 2002 Aug;138(8):1082-6.
414. Toms LM, Allmyr M, Mueller JF, Adolfsson-Erici M, McLachlan M, Murby J, Harden FA. Triclosan in individual human milk samples from Australia. Chemosphere. 2011 Dec;85(11):1682-6. doi: 10.1016/j.chemosphere.2011.08.009. Epub 2011 Oct 13.
415. Adolfsson-Erici M, Pettersson M, Parkkonen J, Sturve J. Triclosan, a commonly used bactericide found in human milk and in the aquatic environment in Sweden. Chemosphere. 2002 Mar;46(9-10):1485-9.
416. Allmyr M, Adolfsson-Erici M, McLachlan MS, Sandborgh-Englund G. Triclosan in plasma and milk from Swedish nursing mothers and their exposure via personal care products. Sci Total Environ. 2006 Dec 15;372(1):87-93. Epub 2006 Sep 26.
417. Allmyr M, McLachlan MS, Sandborgh-Englund G, Adolfsson-Erici M. Determination of triclosan as its pentafluorobenzoyl ester in human plasma and milk using electron capture negative ionization mass spectrometry. Anal Chem. 2006 Sep 15;78(18):6542-6.
418. Dayan AD. Risk assessment of triclosan [Irgasan] in human breast milk. Food Chem Toxicol. 2007 Jan;45(1):125-9. Epub 2006 Aug 30.
419. Calafat AM, Ye X, Wong LY, Reidy JA, Needham LL. Urinary concentrations of triclosan in the U.S. population: 2003-2004. Environ Health Perspect. 2008 Mar;116(3):303-7. doi: 10.1289/ehp.10768.
420. Wolff MS, Teitelbaum SL, Pinney SM, Windham G, Liao L, Biro F, Kushi LH, Erdmann C, Hiatt RA, Rybak ME, Calafat AM. Investigation of relationships between urinary biomarkers of phytoestrogens, phthalates, and phenols and pubertal stages in girls. Breast Cancer and Environment Research Centers. Environ Health Perspect. 2010 Jul;118(7):1039-46. doi: 10.1289/ehp.0901690. Epub 2010 Mar 22.
421. Bertelsen RJ, Engel SM, Jusko TA, Calafat AM, Hoppin JA, London SJ, Eggesbø M, Aase H, Zeiner P, Reichborn-Kjennerud T, Knudsen GP, Guidry VT, Longnecker MP. Reliability of triclosan measures in repeated urine samples from Norwegian pregnant women. J Expo Sci Environ Epidemiol. 2014 Jan 29. doi: 10.1038/jes.2013.95. [Epub ahead of print]
422. Kolpin DW, Furlong ET, Meyer MT, Thurman EM, Zaugg SD, Barber LB, Buxton HT. Pharmaceuticals, hormones, and other organic wastewater contaminants in U.S. streams, 1999-2000: a national reconnaissance. Environ Sci Technol. 2002 Mar 15;36(6):1202-11.
423. McAvoy DC, Schatowitz B, Jacob M, Hauk A, Eckhoff WS. Measurement of triclosan in wastewater treatment systems. Environ Toxicol Chem. 2002 Jul;21(7):1323-9.
424. Kanda R, Griffin P, James HA, Fothergill J. Pharmaceutical and personal care products in sewage treatment works. J Environ Monit. 2003 Oct;5(5):823-30.
425. http://www.ewg.org/research/down-drain/%C2%BB-triclosan
426. Canosa P, Rodríguez I, Rubí E, Cela R. Determination of parabens and triclosan in indoor dust using matrix solid-phase dispersion and gas chromatography with tandem mass spectrometry. Anal Chem. 2007 Feb 15;79(4):1675-81.
427. Calafat AM, Ye X, Wong LY, Reidy JA, Needham LL. Urinary concentrations of triclosan in the U.S. population: 2003-2004. Environ Health Perspect. 2008 Mar;116(3):303-7. doi: 10.1289/ehp.10768.
428. Geens T, Neels H, Covaci A. Distribution of bisphenol-A, triclosan and n-nonylphenol in human adipose tissue, liver and brain. Chemosphere. 2012 May;87(7):796-802. doi: 10.1016/j.chemosphere. 2012.01.002. Epub 2012 Jan 24.
429. Erika S. Koeppe, Kelly K. Ferguson, Justin A. Colacino, John D. Meeker. Relationship between Urinary Triclosan and Paraben Concentrations and Serum Thyroid Measures in NHANES 2007–2008. Sci Total Environ. Author manuscript; available in PMC 2014 February 15. Published in final edited form as: Sci Total Environ. 2013 February 15; 445-446: 299–305. Published online 2013 January 20.
430. Katie B. Paul, Joan M. Hedge, Ruby Bansal, R. Thomas Zoeller, Robert Peter, Michael J. DeVito, Kevin M. Crofton. Developmental Triclosan Exposure Decreases Maternal, Fetal, and Early Neonatal Thyroxine: Dynamic and Kinetic Evaluation of a Putative Mode-of-Action. Toxicology. Author manuscript; available in PMC 2013 October 9. Published in final edited form as: Toxicology. 2012 October 9; 300(1-2): 31–45.
431. Axelstad M, Boberg J, Vinggaard AM, Christiansen S, Hass U. Triclosan exposure reduces thyroxine levels in pregnant and lactating rat dams and in directly exposed offspring. Food Chem Toxicol. 2013 Sep;59:534-40. doi: 10.1016/j.fct.2013.06.050. Epub 2013 Jul 4.
432. Crofton KM, Paul KB, Devito MJ, Hedge JM. Short-term in vivo exposure to the water contaminant triclosan: Evidence for disruption of thyroxine. Environ Toxicol Pharmacol. 2007 Sep;24(2):194-7. doi: 10.1016/j.etap.2007.04.008. Epub 2007 Apr 27.
433. Paul KB, Hedge JM, Devito MJ, Crofton KM. Developmental triclosan exposure decreases maternal and neonatal thyroxine in rats. Environ Toxicol Chem. 2010 Dec;29(12):2840-4. doi: 10.1002/etc.339. Epub 2010 Oct 15.
434. Paul KB, Hedge JM, DeVito MJ, Crofton KM. Short-term exposure to triclosan decreases thyroxine in vivo via upregulation of hepatic catabolism in Young Long-Evans rats. Toxicol Sci. 2010 Feb;113(2):367-79. doi: 10.1093/toxsci/kfp271. Epub 2009 Nov 12.
435. Rodríguez PE1, Sanchez MS. Maternal exposure to triclosan impairs thyroid homeostasis and female pubertal development in Wistar rat offspring. J Toxicol Environ Health A. 2010;73(24):1678-88. doi: 10.1080/15287394.2010.516241.
436. Zorrilla LM, Gibson EK, Jeffay SC, Crofton KM, Setzer WR, Cooper RL, Stoker TE. The effects of triclosan on puberty and thyroid hormones in male Wistar rats. Toxicol Sci. 2009 Jan;107(1):56-64. doi: 10.1093/toxsci/kfn225. Epub 2008 Oct 21.
437. Marlatt VL, Veldhoen N, Lo BP, Bakker D, Rehaume V, Vallée K, Haberl M, Shang D, van Aggelen GC, Skirrow RC, Elphick JR, Helbing CC. Triclosan exposure alters postembryonic development in a Pacific tree frog (Pseudacris regilla) Amphibian Metamorphosis Assay (TREEMA). Aquat Toxicol. 2013 Jan 15;126:85-94. doi: 10.1016/j.aquatox.2012.10.010. Epub 2012 Oct 29.
438. Fort DJ, Mathis MB, Hanson W, Fort CE, Navarro LT, Peter R, Büche C, Unger S, Pawlowski S, Plautz JR. Triclosan and thyroid-mediated metamorphosis in anurans: differentiating growth effects from thyroid-driven metamorphosis in Xenopus laevis. Toxicol Sci. 2011 Jun;121(2):292-302. doi: 10.1093/toxsci/kfr069. Epub 2011 Mar 23.
439. Veldhoen N, Skirrow RC, Osachoff H, Wigmore H, Clapson DJ, Gunderson MP, Van Aggelen G, Helbing CC. The bactericidal agent triclosan modulates thyroid hormone-associated gene expression and disrupts postembryonic anuran development. Aquat Toxicol. 2006 Dec 1;80(3):217-27. Epub 2006 Sep 29. Erratum in: Aquat Toxicol. 2007 Jun 5;83(1):84.
440. Dann AB, Hontela A. Triclosan: environmental exposure, toxicity and mechanisms of action. J Appl Toxicol. 2011 May;31(4):285-311. doi: 10.1002/jat.1660. Review.
441. Ishibashi H, Matsumara N, Hirano M, Matsuoka M, Shiratsuchi H, Ishibashih Y, Takao Y, Arizono K. Effects of triclosan on the early life stages and reproduction of medaka Oryzias latipes and induction of hepatic vitellogenin. Aquat Toxicol. 2004;67:167–79.
442. Raut SA, Angus RA. Triclosan has endocrine-disrupting effects in male western mosquitofish, Gambusia affinis. Environ Toxicol Chem. 2010;29:1287–91.
443. Torres-Duarte C, Viana MT, Vazquez-Duhalt R. Laccase-mediated transformations of endocrine disrupting chemicals abolish binding affinities to estrogen receptors and their estrogenic activity in zebrafish. Appl Biochem Biotechnol. 2012 Oct;168(4):864-76. doi: 10.1007/s12010-012-9825-2. Epub 2012 Sep 2.
444. Forgacs AL, Ding Q, Jaremba RG, Huhtaniemi IT, Rahman NA, Zacharewski TR. BLTK1 murine Leydig cells: a novel steroidogenic model for evaluating the effects of reproductive and developmental toxicants. Toxicol Sci. 2012 Jun;127(2):391-402. doi: 10.1093/toxsci/kfs121. Epub 2012 Mar 29.
445. Kumar V, Chakraborty A, Kural MR, Roy P. Alteration of testicular steroidogenesis and histopathology of reproductive system in male rats treated with triclosan. Reprod Toxicol. 2009 Apr;27(2):177-85. doi: 10.1016/j.reprotox.2008.12.002. Epub 2008 Dec 11.
446. Foran CM, Bennett ER, Genson WH. Developmental evaluation of a potential non-steroidal estrogen: triclosan. Mar Environ Res. 2000;50:153–6.
447. Gee RH, Charles A, Taylor N, Darbre PD. Oestrogenic and androgenic activity of triclosan in breast cancer cells. J Appl Toxicol. 2008 Jan;28(1):78-91.
448. Felicia Udoji, Tamara Martin, Rachel Etherton, Margaret M. Whalen. Immunosuppressive Effects of Triclosan, Nonylphenol, and DDT on Human Natural Killer Cells In Vitro. J Immunotoxicol. Author manuscript; available in PMC 2011 July 1.
449. Sicherer SH, Leung DY. Advances in allergic skin disease, anaphylaxis, and hypersensitivity reactions to foods, drugs, and insects in 2012. J Allergy Clin Immunol. 2013 Jan;131(1):55-66. doi: 10.1016/j.jaci.2012.11. 007. Epub 2012 Nov 27. Review.
450. Michael T. Dinwiddie, Paul D. Terry, Jiangang Chen. Recent Evidence Regarding Triclosan and Cancer Risk. Int J Environ Res Public Health. 2014 February; 11(2): 2209–2217. Published online 2014 February 21. doi: 10.3390/ijerph110202209
451. Ahn K.C., Zhao B., Chen J., Cherednichenko G., Sanmarti E., Denison M.S., Lasley B., Pessah I.N., Kultz D., Chang D.P., Gee S.J., Hammock B.D. In vitro biologic activities of the antimicrobials triclocarban, its analogs, and triclosan in bioassay screens: Receptor-based bioassay screens. Environ. Health Perspect. 2008;116:1203–1210. doi: 10.1289/ehp.11200.
452. Gee R.H., Charles A., Taylor N., Darbre P.D. Oestrogenic and androgenic activity of triclosan in breast cancer cells. J. Appl. Toxicol. 2008;28:78–91. doi: 10.1002/jat.1316.
453. Henry N.D., Fair P.A. Comparison of in vitro cytotoxicity, estrogenicity and anti-estrogenicity of triclosan, perfluorooctane sulfonate and perfluorooctanoic acid. J. Appl. Toxicol. 2013;33:265–272. doi: 10.1002/jat.1736.
454. Gee R.H., Charles A., Taylor N., Darbre P.D. Oestrogenic and androgenic activity of triclosan in breast cancer cells. J. Appl. Toxicol. 2008;28:78–91. doi: 10.1002/jat.1316.
455. Darbre P.D. Environmental oestrogens, cosmetics and breast cancer. Best Pract. Res. Clin. Endocrinol. Metab. 2006;20:121–143. doi: 10.1016/j.beem.2005.09.007.
456. Recchia A.G., Vivacqua A., Gabriele S., Carpino A., Fasanella G., Rago V., Bonofiglio D., Maggiolini M. Xenoestrogens and the induction of proliferative effects in breast cancer cells via direct activation of oestrogen receptor alpha. Food Add. Contam. 2004;21:134–144. doi: 10.1080/02652030310001641177.
457. Michael T. Dinwiddie, Paul D. Terry, Jiangang Chen. Recent Evidence Regarding Triclosan and Cancer Risk. Int J Environ Res Public Health. 2014 February; 11(2): 2209–2217. Published online 2014 February 21. doi: 10.3390/ijerph110202209
458. Rodricks J.V., Swenberg J.A., Borzelleca J.F., Maronpot R.R., Shipp A.M. Triclosan: A critical review of the experimental data and development of margins of safety for consumer products. Crit. Rev. Toxicol. 2010;40:422–484. doi: 10.3109/10408441003667514.
459. Michael T. Dinwiddie, Paul D. Terry, Jiangang Chen. Recent Evidence Regarding Triclosan and Cancer Risk. Int J Environ Res Public Health. 2014 February; 11(2): 2209–2217. Published online 2014 February 21. doi: 10.3390/ijerph110202209
460. Catherine m. Cooney. Personal care products: triclosan comes under scrutiny. Environ health perspect. 2010 june; 118(6): a242. Doi: 10.1289/ehp.118-a242
461. Fiss EM, et al. Environ Sci Technol. 2007;41(7):2387–2394.
462. Lores M1, Llompart M, Sanchez-Prado L, Garcia-Jares C, Cela R. Confirmation of the formation of dichlorodibenzo-p-dioxin in the photodegradation of triclosan by photo-SPME. Anal Bioanal Chem. 2005 Mar;381(6):1294-8. Epub 2005 Feb 9.