The Untold Perils of Proton Pump Inhibitors (PPIs)
As we learned in the last article, gastroesophageal reflux disease (GERD) is an extremely prevalent condition in our society. It has been estimated the prevalence of GERD symptoms in the general population may be up to 30% in the U.S.[125] but other studies show it could be as high as 34% to 40%.[126] The standard treatment for this condition continues to be the use of a powerful class of medications known as proton-pump inhibitors (PPIs). PPIs are block-buster drugs that make the pharmaceutical companies billions of dollars annually. This class of medication represents the third-highest selling class of drugs in the United States, with $13.9 billion in annual sales.[127] Sales of Nexium (the “purple pill” by Astra Zeneca) topped $6.1 billion in 2013 alone, the second highest prescribed medication in the U.S.[128] In this article we take a closer look at the known risks associated with the use of PPIs, according to the most recent literature.
 PPIs and Nutrient Absorption
PPIs and Nutrient Absorption
Since stomach acid plays a role in nutrient absorption, PPIs and other acid suppressing medications have a significant impact on vitamin and mineral absorption. The literature reveals that absorption of various vitamins and minerals are impacted as a result of acid suppression.
 PPIs and Vitamin B12 Absorption
PPIs and Vitamin B12 Absorption
Vitamin B12, also called cobalamin, is a crucial water-soluble vitamin that is used for many functions in the body, including brain function, nerve function, red blood cell production, methylation and liver detoxification. B12 deficiency is associated with a number of chronic diseases, including heart disease and cancer. Our primary source of B12 is our diet, and it is found in animal products such as meat. However, in its dietary form, B12 is bound to animal-derived protein which requires stomach acid to cleave off the protein so that it can be absorbed after it becomes bound to intrinsic factor in the intestines. 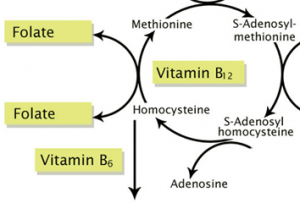 If stomach acid is insufficient, B12 may not be cleaved off from dietary proteins and lead to absorption problems.
If stomach acid is insufficient, B12 may not be cleaved off from dietary proteins and lead to absorption problems.
“The use of H(2)-receptor antagonists and/or PPIs may impair the absorption of protein-bound dietary vitamin B12 and could contribute to the development of vitamin B12 deficiency with prolonged use.”[189]
“The studies reviewed in this article clearly show that omeprazole therapy will decrease the absorption of vitamin B12 by preventing its cleavage from dietary proteins.”[190]
 A number of studies have shown an association of decreased B12 absorption with prolonged PPI use.[189,190] However, PPIs have been shown to affect B12 absorption in very short periods of time as well.[191,192] One study showed a 72% decrease in B12 absorption on 20 mg of omeprazole (Prilosec) and an 88% decrease in B12 absorption on 40 mg of omeprazole after only 2 weeks of treatment! The authors made the following conclusion:
A number of studies have shown an association of decreased B12 absorption with prolonged PPI use.[189,190] However, PPIs have been shown to affect B12 absorption in very short periods of time as well.[191,192] One study showed a 72% decrease in B12 absorption on 20 mg of omeprazole (Prilosec) and an 88% decrease in B12 absorption on 40 mg of omeprazole after only 2 weeks of treatment! The authors made the following conclusion:
“Omeprazole (PPI) therapy acutely decreased cyanocobalamin (B12) absorption in a dose-dependent manner.”[191]
 B12 absorption issues worsen in patients with atrophic gastritis, which is a common condition in the elderly. In one study of 359 elderly people aged between 60 and 99 years of age, the prevalence of atrophic gastritis was found to be 31.5% and increased significantly with advancing age. Subjects with atrophic gastritis exhibited a lower mean serum vitamin B12 level, indicating decreased absorption.
B12 absorption issues worsen in patients with atrophic gastritis, which is a common condition in the elderly. In one study of 359 elderly people aged between 60 and 99 years of age, the prevalence of atrophic gastritis was found to be 31.5% and increased significantly with advancing age. Subjects with atrophic gastritis exhibited a lower mean serum vitamin B12 level, indicating decreased absorption.
“It is concluded that…atrophic gastritis is common in an elderly population, and that atrophic gastritis is associated with vitamin B12 deficiency and anemia.”[193]
 PPIs and Iron Absorption
PPIs and Iron Absorption
Iron is an essential element for red blood cell production. About 70% of your body’s iron is found in the red blood cells of your blood called hemoglobin and in muscle cells called myoglobin. Hemoglobin is essential for transferring oxygen in your blood from the lungs to the tissues. It has long been known that conditions of low stomach acid (hypochlorhydria) such as atrophic gastritis and partial gastrectomy cause iron-deficiency anemia.[194] Studies have shown that patients who undergo gastric bypass surgeries are at much higher risk of iron deficiency, in fact, iron deficiency is extremely frequent in this patient population.[195]
 Iron can be found in the heme (ferrous) form or nonheme form but only the ferrous form can be absorbed. Nonheme iron requires gastric acid for conversion to the ferrous form of iron for absorption.[196] 90% of dietary and 100% of oral iron supple-mentation is in the nonheme form.[197] So not surprisingly, several review studies in the literature conclude that PPI use leads to decreased iron absorption.[198,199] In one study, all markers of iron deficiency were decreased among patients on PPI therapy as compared to controls.
Iron can be found in the heme (ferrous) form or nonheme form but only the ferrous form can be absorbed. Nonheme iron requires gastric acid for conversion to the ferrous form of iron for absorption.[196] 90% of dietary and 100% of oral iron supple-mentation is in the nonheme form.[197] So not surprisingly, several review studies in the literature conclude that PPI use leads to decreased iron absorption.[198,199] In one study, all markers of iron deficiency were decreased among patients on PPI therapy as compared to controls.
 “Among patients on PPI therapy, all hematologic indices decreased from baseline, including hemoglobin (-0.19 g/dL, P=0.03), hematocrit (-0.63%, P=0.02), and mean corpuscular volume (-0.49 fL, P=0.05). PPI users had significant decreases in mean hemoglobin and hematocrit (P<0.01 for both) compared with matched controls. Among adult patients receiving chronic PPI therapy, there is a significant decrease in hematologic indices from baseline.”[200]
“Among patients on PPI therapy, all hematologic indices decreased from baseline, including hemoglobin (-0.19 g/dL, P=0.03), hematocrit (-0.63%, P=0.02), and mean corpuscular volume (-0.49 fL, P=0.05). PPI users had significant decreases in mean hemoglobin and hematocrit (P<0.01 for both) compared with matched controls. Among adult patients receiving chronic PPI therapy, there is a significant decrease in hematologic indices from baseline.”[200]
Another study looked at the effects of omeprazole on iron supplementation in patients with iron-deficient anemia. It found that:
 “…the profound hypochlorhydria induced by omeprazole may indeed impair the optimal absorption of orally administered iron in iron-deficient individuals…”[201]
“…the profound hypochlorhydria induced by omeprazole may indeed impair the optimal absorption of orally administered iron in iron-deficient individuals…”[201]
In fact, PPI therapy is so reliable in reducing iron absorption from food, it is now being used as a treatment for decreasing iron absorption from food in conditions of excess iron stores in the body called hemochromatosis.
“Proton pump inhibitor therapy reduces the absorption of non-heme iron and this effect has been employed in the management of hemochromatosis. It may also retard clinical response to iron supplementation.”[202]
 PPIs and Calcium Absorption
PPIs and Calcium Absorption
Calcium performs a number of basic functions in your body. Your body uses 99% of its calcium to keep your bones and teeth strong, thereby supporting skeletal structure and function. The rest of the calcium in your body plays key roles in cell signaling, blood clotting, muscle contraction and nerve function but calcium levels for these functions is usually tightly-regulated. So anything that effects calcium absorption from food will likely have a negative impact on bone density, primarily. Numerous human studies show that gastric acid secretion aid in calcium absorption and that acid suppressants such as PPIs can decrease calcium absorption and decrease bone density.[203-210] An acidic environment in the stomach facilitates the release of ionized calcium from calcium salts, which is important for calcium absorption.[203-210] A number of conditions that cause hypochlorhydria are associated with an increased occurrence of osteoporosis and bone fracture, and it is assumed that this is secondary to the effect of low gastric acid levels on calcium absorption.[203-210]
 There were two large, case control studies from 2006 which led to considerable concern that chronic PPI use could lead to an increase in bone fractures and speculation of the possible mechanisms involved. Both of these studies associated long-term use of PPIs with bone fractures with one of them showing the risks were particularly increased with high-dose PPI use and the risk progressively increased with the duration of PPI treatment.[205,211]
There were two large, case control studies from 2006 which led to considerable concern that chronic PPI use could lead to an increase in bone fractures and speculation of the possible mechanisms involved. Both of these studies associated long-term use of PPIs with bone fractures with one of them showing the risks were particularly increased with high-dose PPI use and the risk progressively increased with the duration of PPI treatment.[205,211]
These studies and others led the U.S. Food and Drug Administration to issue a warning in 2010 of the “possible increased risk of fractures of the hip, wrist, and spine with high doses or long-term use of a class of medications called proton pump inhibitors.” (U.S. FDA News Release, May 25, 2010).  Since then, PPIs and other acid-suppressing medications have been shown in numerous studies to decrease calcium absorption [208, 212-214] with omeprazole causing a 41% decrease in one study [208] and long-term PPI usage has been increasingly associated with an increased occurrence of bone fractures. [210,211,215-217] The most widely-assumed mechanism is that long-term PP
Since then, PPIs and other acid-suppressing medications have been shown in numerous studies to decrease calcium absorption [208, 212-214] with omeprazole causing a 41% decrease in one study [208] and long-term PPI usage has been increasingly associated with an increased occurrence of bone fractures. [210,211,215-217] The most widely-assumed mechanism is that long-term PP I use leads to decreased intestinal absorption of calcium resulting in increased osteoporosis, increased bone loss and increased fractures. [205,206, 210,212,218-220]
I use leads to decreased intestinal absorption of calcium resulting in increased osteoporosis, increased bone loss and increased fractures. [205,206, 210,212,218-220]
A large retrospective analysis done in 2008 matched 15,792 cases of osteoporosis-related fractures with 47,289 controls during an 8 year period between 1996 and 2004. The researchers concluded the following:
“Use of proton pump inhibitors for 7 or more years is associated with a significantly increased risk of an osteoporosis-related fracture. There is an increased risk of hip fracture after 5 or more years exposure.”[221]
 PPIs and Magnesium Absorption
PPIs and Magnesium Absorption
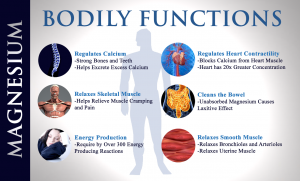
Magnesium is needed for more than 300 biochemical reactions in the body. It helps to maintain normal nerve and muscle function, supports a healthy immune system, keeps the heart beat steady, and helps bones remain strong. It also helps regulate blood glucose levels and aids in the production of energy and protein. So anything that impacts magnesium, can potentially cause a wide range of symptoms related to the muscles, nerves, brain, heart, bone and the immune system.
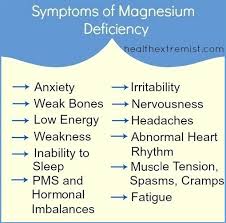
 Hypomagnesemia (low magnesium levels in the blood) was reported for the first time in the medical literature as a potential side effect of proton-pump inhibitors (PPIs) in 2006. Since then, numerous studies have shown that magnesium deficiency associated with the use of PPIs results in a wide variety of serious disorders affecting multiple organ systems.[222-229] In 2011, the U.S. Food and Drug Administration released a warning about low serum magnesium levels associated with long-term use of PPIs. Symptoms include seizures, arrhythmias, low blood pressure, severe muscle contraction (tetany), and death.[230]
Hypomagnesemia (low magnesium levels in the blood) was reported for the first time in the medical literature as a potential side effect of proton-pump inhibitors (PPIs) in 2006. Since then, numerous studies have shown that magnesium deficiency associated with the use of PPIs results in a wide variety of serious disorders affecting multiple organ systems.[222-229] In 2011, the U.S. Food and Drug Administration released a warning about low serum magnesium levels associated with long-term use of PPIs. Symptoms include seizures, arrhythmias, low blood pressure, severe muscle contraction (tetany), and death.[230]
 In a recent review of 10 cases, the elderly patients had been taking PPIs for an average of 8.3 yrs.[226] They suffered from severe symptoms of low magnesium and there was significant morbidity related to fatigue, unsteadiness, severe muscle contraction, seizures, cardiac arrhythmias, and multiple hospitalizations. The low magnesium condition promptly resolved when the PPI therapy was stopped and recurred if the PPI therapy was reintroduced.
In a recent review of 10 cases, the elderly patients had been taking PPIs for an average of 8.3 yrs.[226] They suffered from severe symptoms of low magnesium and there was significant morbidity related to fatigue, unsteadiness, severe muscle contraction, seizures, cardiac arrhythmias, and multiple hospitalizations. The low magnesium condition promptly resolved when the PPI therapy was stopped and recurred if the PPI therapy was reintroduced.
“There were 18 emergency hospital admissions with severe hypomagnesemia (low magnesium). Oral and parenteral magnesium supplements were relatively ineffective at correcting the problem, but stopping PPI therapy lead to prompt resolution of the hypomagnesemia (within 2 weeks in five carefully monitored patients), with symptomatic benefit. Hypomagnesemia recurred if PPI therapy was re-introduced…”[226]
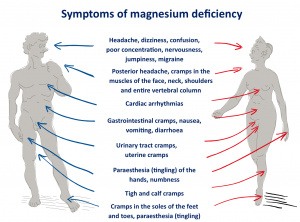 Another recent review of 4 cases examined the clinical consequences and mechanisms of electrolyte disorders that resulted from the elderly patients taking a PPI for a period of 1 to 13 years.[227] They developed severe low magnesium levels with low calcium levels, hypoparathyroidism (low parathyroid hormone), and extremely low urinary calcium and magnesium excretion. One patient developed encephalopathy (brain dysfunction) after a collapse likely caused by cardiac arrhythmia. The others had cardiac abnormalities identified by electrocardiogram. Low potassium levels were considered the cause for these arrhythmias. The low potassium levels were believed to be due to excess potassium excretion by the body resulting from the low magnesium.
Another recent review of 4 cases examined the clinical consequences and mechanisms of electrolyte disorders that resulted from the elderly patients taking a PPI for a period of 1 to 13 years.[227] They developed severe low magnesium levels with low calcium levels, hypoparathyroidism (low parathyroid hormone), and extremely low urinary calcium and magnesium excretion. One patient developed encephalopathy (brain dysfunction) after a collapse likely caused by cardiac arrhythmia. The others had cardiac abnormalities identified by electrocardiogram. Low potassium levels were considered the cause for these arrhythmias. The low potassium levels were believed to be due to excess potassium excretion by the body resulting from the low magnesium.
 “This series of PPI-induced hypomagnesemia shows that this is a generic effect. It also indicates that hypomagnesemia may occur within 1 year of PPI therapy initiation and can have serious clinical consequences…”[227]
“This series of PPI-induced hypomagnesemia shows that this is a generic effect. It also indicates that hypomagnesemia may occur within 1 year of PPI therapy initiation and can have serious clinical consequences…”[227]
At present the mechanism of the PPI-induced low magnesium levels is not known. It was concluded in one study that it is not specific to a given PPI, but is a generic problem with the PPI class of drugs, because it recurs even when PPIs are interchanged.[82] It is believed that low magnesium levels resulting from PPI use is likely due to gastrointestinal malabsorption of magnesium secondary to hypochlorhydria.[222]
 Summary of PPIs and Nutrient Absorption
Summary of PPIs and Nutrient Absorption
As you can see, the evidence supporting the risk of critical vitamin and mineral malabsorption secondary to acid suppression by PPI use is alarming and unfortunately often overlooked by doctors and patients. In addition to risk of vitamin B12, calcium, iron and magnesium malabsorption, there is evidence that acid suppression by PPIs increases risk of malabsorption of other important vitamins and minerals as well, including vitamin C, zinc and folic acid.[231- 235]
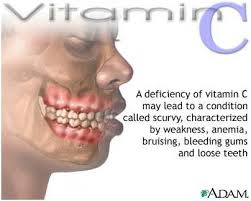 “Proton pump inhibitor therapy lowers the concentration of vitamin C in gastric juice and the proportion of the vitamin in its active antioxidant form i.e., ascorbic acid. There is also some evidence that proton pump inhibitors may reduce the bioavailability of ingested vitamin C.” [231]
“Proton pump inhibitor therapy lowers the concentration of vitamin C in gastric juice and the proportion of the vitamin in its active antioxidant form i.e., ascorbic acid. There is also some evidence that proton pump inhibitors may reduce the bioavailability of ingested vitamin C.” [231]
“Zinc absorption decreased (about 42%) after omeprazole (Prilosec) administration compared with pre-omeprazole values. Suppression of gastric acid secretion by omeprazole reduces intestinal absorption of zinc.”[232]
 “This study indicates that intragastric pH and salt solubility-dissolution are important in the oral absorption of zinc.”[233]
“This study indicates that intragastric pH and salt solubility-dissolution are important in the oral absorption of zinc.”[233]
“Further, in vitro studies have confirmed the dependency of folate absorption on intraluminal pH.”[235]
In one study, when folate was given to achlorhydric patients (with no stomach acid) along with an HCl supplement, absorption of folate increased by 54%.[235] Although there is currently no available research on PPI risk of malabsorption of other nutrients, given that gastrointestinal pH plays an important role in absorption of various vitamins and minerals, one has to consider the probability that the absorption of other nutrients are affected as well.
 PPIs and SIBO
PPIs and SIBO
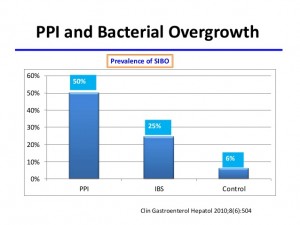
Small intestinal bacterial overgrowth (SIBO) commonly occurs in patients on omeprazole (PPI) and in atrophic gastritis (a condition leading to very low stomach acid).[183]
“SIBO…occurs significantly more frequently among long term PPI users than patients with IBS or control subjects.”[184]
 Considering that low stomach acid is an important risk factor for the development of SIBO, recent research has focused on the mechanism by which PPIs lead to SIBO. The mechanism appears to be related to a shift in the balance of the intestinal bacteria that occurs in PPI use. This should not be surprising, given the important role that acid plays in preventing bacterial infection.
Considering that low stomach acid is an important risk factor for the development of SIBO, recent research has focused on the mechanism by which PPIs lead to SIBO. The mechanism appears to be related to a shift in the balance of the intestinal bacteria that occurs in PPI use. This should not be surprising, given the important role that acid plays in preventing bacterial infection.
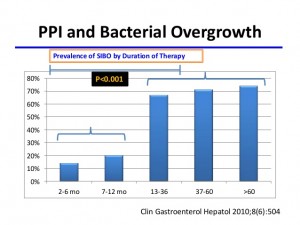 “Use of proton pump inhibitors (PPIs) could predispose individuals to small intestinal bacterial overgrowth (SIBO) by altering the intraluminal environment and bacterial flora.”[185]
“Use of proton pump inhibitors (PPIs) could predispose individuals to small intestinal bacterial overgrowth (SIBO) by altering the intraluminal environment and bacterial flora.”[185]
“In conclusion, omeprazole is associated with a microbiota shift and functional change in the distal gut in patients with compensated cirrhosis that could set the stage for bacterial overgrowth.”[186]
 PPIs and Colonic Infections
PPIs and Colonic Infections
PPIs have also been shown to increase risk of Clostridium difficile (known as C. diff) infection, which is commonly seen in patients on long-term antibiotic therapy.
“Omeprazole treatment decreases the expression of genes that have important functions in colonocyte integrity. Such impairment in colonocyte function may promote Clostridium difficile infection (CDI).”[236]
 PPIs and Immune Responses
PPIs and Immune Responses
PPIs have also been shown to affect normal immune responses and clotting mechanisms.
“One potential toxicity mechanism of PPIs as suggested from this systematic PPI-specific gene expression analysis is that PPIs are enriched in acidic organelles where they are activated and inhibit V-ATPases and acid hydrolases, and consequently block the pathways of antigen presentation, the synthesis and secretion of cytokines, and complement component proteins and coagulation factors.”[237]
Related Articles
Hypochlorhydria (Low Stomach Acid)
Gastroesophageal Reflux Disease (GERD)
Intestinal Permeability/Leaky Gut
Irritable Bowel Syndrome (IBS)
Small Intestinal Bacterial Overgrowth
The Neuroendocrine System of the Gut and the Brain-Gut Axis
References
189. Ruscin JM1, Page RL 2nd, Valuck RJ. Vitamin B(12) deficiency associated with histamine(2)-receptor antagonists and a proton-pump inhibitor. Ann Pharmacother. 2002 May;36(5):812-6.
190. Bradford GS1, Taylor CT. Omeprazole and vitamin B12 deficiency. Ann Pharmacother. 1999 May;33(5):641-3.
191. Marcuard SP1, Albernaz L, Khazanie PG. Omeprazole therapy causes malabsorption of cyanocobalamin (vitamin B12). Ann Intern Med. 1994 Feb 1;120(3):211-5.
192. Schenk BE1, Festen HP, Kuipers EJ, Klinkenberg-Knol EC, Meuwissen SG. Effect of short- and long-term treatment with omeprazole on the absorption and serum levels of cobalamin. Aliment Pharmacol Ther. 1996 Aug;10(4):541-5.
193. Krasinski SD, Russell RM, Samloff IM, Jacob RA, Dallal GE, McGandy RB, Hartz SC. Fundic atrophic gastritis in an elderly population. Effect on hemoglobin and several serum nutritional indicators. J Am Geriatr Soc. 1986 Nov;34(11):800-6.
194. Annibale B, Capurso G, Delle Fave G. The stomach and iron deficiency anaemia: a forgotten link. Dig Liver Dis. 2003;35:288–295.
195. Gesquiere I, Lannoo M, Augustijns P, Matthys C, Van der Schueren B, Foulon V.
Iron deficiency after Roux-en-Y gastric bypass: insufficient iron absorption from oral ironsupplements. J Lab Clin Med. 1978 Jul;92(1):108-16.
196. Bezwoda W, Charlton R, Bothwell T, Torrance J, Mayet F. The importance of gastric hydrochloric acid in the absorption of nonheme food iron. Obes Surg. 2014 Jan;24(1):56-61. doi: 10.1007/s11695-013-1042-8.
197. Tempel M, Chawla A, Messina C, Celiker MY. Effects of omeprazole on iron absorption: preliminary study. Turk J Haematol. 2013 Sep;30(3):307-10. doi: 10.5152/tjh.2013.0042. Epub 2013 Sep 5.
198. Wilhelm SM, Rjater RG, Kale-Pradhan PB. Perils and pitfalls of long-term effects of proton pump inhibitors. Expert Rev Clin Pharmacol. 2013 Jul;6(4):443-51. doi: 10.1586/17512433.2013.811206. Review.
199. Ito T, Jensen RT. Association of long-term proton pump inhibitor therapy with bone fractures and effects onabsorption of calcium, vitamin B12, iron, and magnesium. Curr Gastroenterol Rep. 2010 Dec;12(6):448-57. doi: 10.1007/s11894-010-0141-0. Review.
200. Sarzynski E, Puttarajappa C, Xie Y, Grover M, Laird-Fick H. Association between proton pump inhibitor use and anemia: a retrospective cohort study. Dig Dis Sci. 2011 Aug;56(8):2349-53. doi: 10.1007/s10620-011-1589-y. Epub 2011 Feb 12.
201. Sharma VR, Brannon MA, Carloss EA. Effect of omeprazole on oral iron replacement in patients with iron deficiency anemia. South Med J. 2004 Sep;97(9):887-9.
202. McColl KE. Effect of proton pump inhibitors on vitamins and iron. Am J Gastroenterol. 2009 Mar;104 Suppl 2:S5-9. doi: 10.1038/ajg.2009.45.
203. Tetsuhide Ito, Robert T. Jensen. Association of Long-term Proton Pump Inhibitor Therapy with Bone Fractures and effects on Absorption of Calcium, Vitamin B12, Iron, and Magnesium. Curr Gastroenterol Rep. 2010 December; 12(6): 448–457.
204. Lodato F, Azzaroli F, Turco L, et al. Adverse effects of proton pump inhibitors. Best Pract Res Clin Gastroenterol. 2010;24:193–201. [PubMed]
205. Yang YX, Lewis JD, Epstein S, Metz DC. Long-term proton pump inhibitor therapy and risk of hip fracture. JAMA. 2006;296:2947–2953. [PubMed]
206. Insogna KL. The effect of proton pump-inhibiting drugs on mineral metabolism. Am J Gastroenterol. 2009;104 (Suppl 2):S2–S4. [PubMed]
207. Fournier MR, Targownik LE, Leslie WD. Proton pump inhibitors, osteoporosis, and osteoporosis-related fractures. Maturitas. 2009;64:9–13. [PubMed]
208. O’Connell MB, Madden DM, Murray AM, et al. Effects of proton pump inhibitors on calcium carbonate absorption in women: a randomized crossover trial. Am J Med. 2005;118:778–781. [PubMed]
209. Sipponen P, Harkonen M. Hypochlorhydric stomach: a risk condition for calcium malabsorption and osteoporosis? Scand J Gastroenterol. 2010;45:133–138. [PubMed]
210. Yu EW, Blackwell T, Ensrud KE, et al. Acid-suppressive medications and risk of bone loss and fracture in older adults. Calcif Tissue Int. 2008;83:251–259. [PMC free article] [PubMed]
211. Vestergaard P, Rejnmark L, Mosekilde L. Proton pump inhibitors, histamine H2 receptor antagonists, and other antacid medications and the risk of fracture. Calcif Tissue Int. 2006;79:76–83.
212. Wright MJ, Proctor DD, Insogna KL, Kerstetter JE. Proton pump-inhibiting drugs, calcium homeostasis, and bone health. Nutr Rev. 2008;66:103–108. [PubMed]
213. Recker RR. Calcium absorption and achlorhydria. N Engl J Med. 1985;313:70–73. [PubMed]
214. Graziani G, Como G, Badalamenti S, et al. Effect of gastric acid secretion on intestinal phosphate and calcium absorption in normal subjects. Nephrol Dial Transplant. 1995;10: 1376–1380. [PubMed]
215. Gray SL, LaCroix AZ, Larson J, et al. Proton pump inhibitor use, hip fracture, and change in bone mineral density in postmenopausal women: results from the Women’s Health Initiative. Arch Intern Med. 2010;170:765–771. [PMC free article] [PubMed]
216. Johnson DA. Safety of proton pump inhibitors: current evidence for osteoporosis and interaction with antiplatelet agents. Curr Gastroenterol Rep. 2010;12:167–174. [PubMed]
217. Corley DA. Proton pump inhibitor, H2 antagonists, and risk of hip fracture: a large population-based study. Gastroenterology. 2009;136:A70.
218. Moayyedi P, Cranney A. Hip fracture and proton pump inhibitor therapy: balancing the evidence for benefit and harm. Am J Gastroenterol. 2008;103:2428–2431. [PubMed]
219. Targownik LE, Lix LM, Leung S, Leslie WD. Proton-pump inhibitor use is not associated with osteoporosis or accelerated bone mineral density loss. Gastroenterology. 2010;138:896–904. [PubMed]
220. Laine L. Proton pump inhibitors and bone fractures? Am J Gastroenterol. 2009;104: S21–S26. [PubMed]
221. Laura E. Targownik, Lisa M. Lix, Colleen J. Metge, Heather J. Prior, Stella Leung, William D. Leslie. Use of proton pump inhibitors and risk of osteoporosis-related fractures. CMAJ. 2008 August 12; 179(4): 319–326. doi: 10.1503/cmaj.071330
222. Tetsuhide Ito, Robert T. Jensen. Association of Long-term Proton Pump Inhibitor Therapy with Bone Fractures and effects on Absorption of Calcium, Vitamin B12, Iron, and Magnesium. Curr Gastroenterol Rep. 2010 December; 12(6): 448–457.
223. Cundy T, Dissanayake A. Severe hypomagnesaemia in long-term users of proton-pump inhibitors. Clin Endocrinol (Oxf). 2008;69:338–341. [PubMed]
224. Kuipers MT, Thang HD, Arntzenius AB. Hypomagnesaemia due to use of proton pump inhibitors–a review. Neth J Med. 2009;67:169–172. [PubMed]
225. Shabajee N, Lamb EJ, Sturgess I, Sumathipala RW. Omeprazole and refractory hypomagnesaemia. BMJ. 2008;337:a425. [PMC free article] [PubMed]
226. Mackay JD, Bladon PT. Hypomagnesaemia due to proton-pump inhibitor therapy: a clinical case series. QJM. 2010;103:387–395. A recent report of the characteristic of 10 cases of PPI induce hypomagnesemia emphasizing the refractoriness of it, severity of its manifestations, and disappearance when the PPI is stopped. [PubMed]
227. Hoorn EJ, van der Hoek J, de Man RA, et al. A Case Series of Proton Pump Inhibitor-Induced Hypomagnesemia. Am J Kidney Dis. 2010 [PubMed]
228. Francois M, Levy-Bohbot N, Caron J, Durlach V. Chronic use of proton-pump inhibitors associated with giardiasis: A rare cause of hypomagnesemic hypoparathyroidism? Ann Endocrinol (Paris). 2008;69:446–448.[PubMed]
229. Broeren MA, Geerdink EA, Vader HL, van den Wall Bake AW. Hypomagnesemia induced by several proton-pump inhibitors. Ann Intern Med. 2009;151:755–756. [PubMed]
230. Nand B, Bhagat M. Serious and commonly overlooked side effect of prolonged use of PPI. Am J Med. 2014 Sep;127(9):e5. doi: 10.1016/j.amjmed.2014.06.002. Epub 2014 Jun 10
231. McColl KE. Effect of proton pump inhibitors on vitamins and iron. Am J Gastroenterol. 2009 Mar;104 Suppl 2:S5-9. doi: 10.1038/ajg.2009.45.
232. Ozutemiz AO, Aydin HH, Isler M, Celik HA, Batur Y. Effect of omeprazole on plasma zinc levels after oral zinc administration. Indian J Gastroenterol. 2002 Nov-Dec;21(6):216-8.
233. Henderson LM1, Brewer GJ, et al. Effect of intragastric pH on the absorption of oral zinc acetate and zinc oxide in young healthy volunteers. JPEN J Parenter Enteral Nutr. 1995 Sep-Oct;19(5):393-7.
234. Farrell CP, Morgan M, et al. Proton pump inhibitors interfere with zinc absorption and zinc body stores. Gastroent Res. 2011;4:243–251.
235. Marcos Pedrosa and Robert Russell. Chronic gastritis and hypochlorhydria in the elderly. Ch.11: Folate and vitamin B12 absorption in atrophic gastritis. 1993 by CRC Press, Inc.
183. Saltzman JR1, Kowdley KV, Pedrosa MC, Sepe T, Golner B, Perrone G, Russell RM. Bacterial overgrowth without clinical malabsorption in elderly hypochlorhydric subjects. Gastroenterology. 1994 Mar;106(3):615-23.
184. Lombardo L1, Foti M, Ruggia O, Chiecchio A. Increased incidence of small intestinal bacterial overgrowth during proton pump inhibitor therapy. Clin Gastroenterol Hepatol. 2010 Jun;8(6):504-8. doi: 10.1016/j.cgh.2009.12.022. Epub 2010 Jan 6.
185. Proton pump inhibitor use and the risk of small intestinal bacterial overgrowth: a meta-analysis. Clin Gastroenterol Hepatol. 2013 May;11(5):483-490.
186. Bajaj JS1, Cox IJ2, Betrapally NS3, et al. Systems biology analysis of omeprazole therapy in cirrhosis demonstrates significant shifts in gut microbiota composition and function. Am J Physiol Gastrointest Liver Physiol. 2014 Nov 15;307(10):G951-7. doi:10.1152/ ajpgi.00268. 2014. Epub 2014 Sep 25.
236. Hegarty JP1, Sangster W1, Harris LR 3rd1, Stewart DB2. Proton pump inhibitors induce changes in colonocyte gene expression that may affect Clostridium difficile infection. Surgery. 2014 Oct;156(4):972-8. doi: 10.1016/j.surg.2014.06.074. Epub 2014 Aug 21.
237. Wu D1, Qiu T, Zhang Q, Kang H, Yuan S, Zhu L, Zhu R. Systematic Toxicity Mechanism Analysis of Proton Pump Inhibitors: An In Silico Study. Chem Res Toxicol. 2015 Feb 9. [Epub ahead of print]
