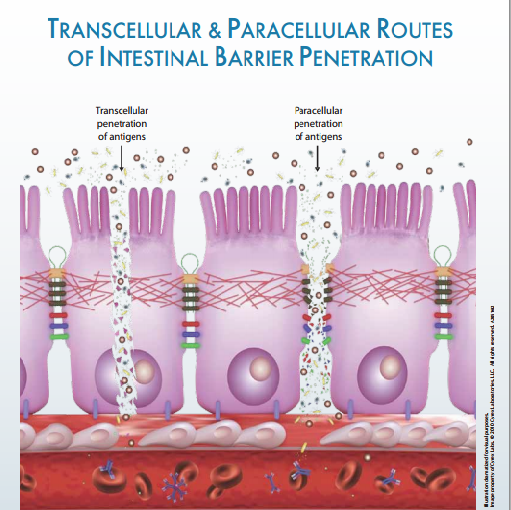
There is extensive research and publications on this condition, including causal factors, associated health conditions and dietary and nutritional compounds that help repair the damage that takes place to the intestinal lining.
There is growing, published evidence showing leaky gut is a precursor for autoimmune diseases, such as type I diabetes, rheumatoid arthritis (RA), lupus (SLE), multiple sclerosis (MS), Lou Gehrig’s disease (ALS), Hashimoto’s hypothyroidism, Grave’s disease, Crohn’s disease, ulcerative colitis, Celiac disease, and the list continues to grow.
Functional Medicine utilizes specialized laboratory testing techniques to identify this condition and determine the extent of the damage to the intestinal lining prior to starting therapy.
Food sensitivity testing may also be used.
Specialized lab monitoring of key markers is used to assess progress throughout the treatment program.
Functional Medicine management of leaky gut involves a restricted dietary regimen avoiding foods that can damage the intestinal lining together with the use of nutritional supplements to help reduce the intestinal inflammation and repair the intestinal lining.
There are a number of nutritional compounds that decrease intestinal inflammation and restore the integrity of the intestinal lining.
A customized dietary and supplementation program is used to overcome a long term degenerative condition.
In almost all cases, a significant improvement, or elimination of symptoms can be achieved.
A restricted whole-food diet that removes common food allergens, inflammatory and processed foods, foods high in sugar content and lectin-containing foods that can damage the intestinal lining is important for a full recovery. A list of foods to consume and to avoid is identified as part of the Functional Medicine management regime.
Other interventions often successfully used include:
Anti-inflammatory and immune-modulating herbs and nutritional compounds to decrease excessive immune responses can very often be helpful.
Milpitas: 408-262-6606
1649 S. Main Street, Ste. 102
Milpitas, CA 95035
Oakland: 510-350-8082
6232 La Salle Ave
Oakland, CA 94611